Coordination Compounds Geometry And Nomenclature

Coordination Compounds Nomenclature Pdf Coordination complexes have their own classes of isomers, different magnetic properties and colors, and various applications (photography, cancer treatment, etc), so it makes sense that they would have a naming system as well. A coordination compound consists of a central metal atom or ion bonded to surrounding molecules or ions called ligands. these bonds are coordinate covalent bonds, meaning the ligand donates both electrons in the shared pair to the metal center.

Coordination Compounds 02 Nomenclature Pdf Ligand Physical In coordination chemistry, compounds can be broadly classified based on their composition and behaviour, particularly in aqueous solutions. two primary types of such compounds are molecular or addition compounds and coordination (or complex) compounds. Werner’s theory the central metal and the ligands directly bonded to it make up the coordination sphere of the complex. in cocl3 ∙ 6 nh3, all six of the ligands are nh3 and the 3 chloride ions are outside the coordination sphere. Complete iupac friendly guide to nomenclature of coordination compounds — rules, examples, mcqs, quiz, faqs, schema markup. A table of the coordination geometries encountered is shown below with examples of their occurrence in complexes found as discrete units in compounds and coordination spheres around atoms in crystals (where there is no discrete complex).

Nomenclature Of Coordination Compounds Pdf Complete iupac friendly guide to nomenclature of coordination compounds — rules, examples, mcqs, quiz, faqs, schema markup. A table of the coordination geometries encountered is shown below with examples of their occurrence in complexes found as discrete units in compounds and coordination spheres around atoms in crystals (where there is no discrete complex). In naming these complexes, cis or trans is written before the names of these compounds. similarly, for octahedral complexes, cis and trans are used in isomerism in coordination compounds. Coordination compounds are named according to the rules suggested by international union of pure and applied chemistry, iupac (1976). these rules are given blow: the positive ion (cation) comes first, followed by the negative ion (anion). this is also the common order for simple salts. Master the systematic naming of coordination compounds with step by step rules, tricks, and jee focused examples. The most common structures of the complexes in coordination compounds are square planar, tetrahedral, and octahedral, corresponding to coordination numbers of four, four, and six, respectively.

Nomenclature Of Coordination Compounds Chemash In naming these complexes, cis or trans is written before the names of these compounds. similarly, for octahedral complexes, cis and trans are used in isomerism in coordination compounds. Coordination compounds are named according to the rules suggested by international union of pure and applied chemistry, iupac (1976). these rules are given blow: the positive ion (cation) comes first, followed by the negative ion (anion). this is also the common order for simple salts. Master the systematic naming of coordination compounds with step by step rules, tricks, and jee focused examples. The most common structures of the complexes in coordination compounds are square planar, tetrahedral, and octahedral, corresponding to coordination numbers of four, four, and six, respectively.
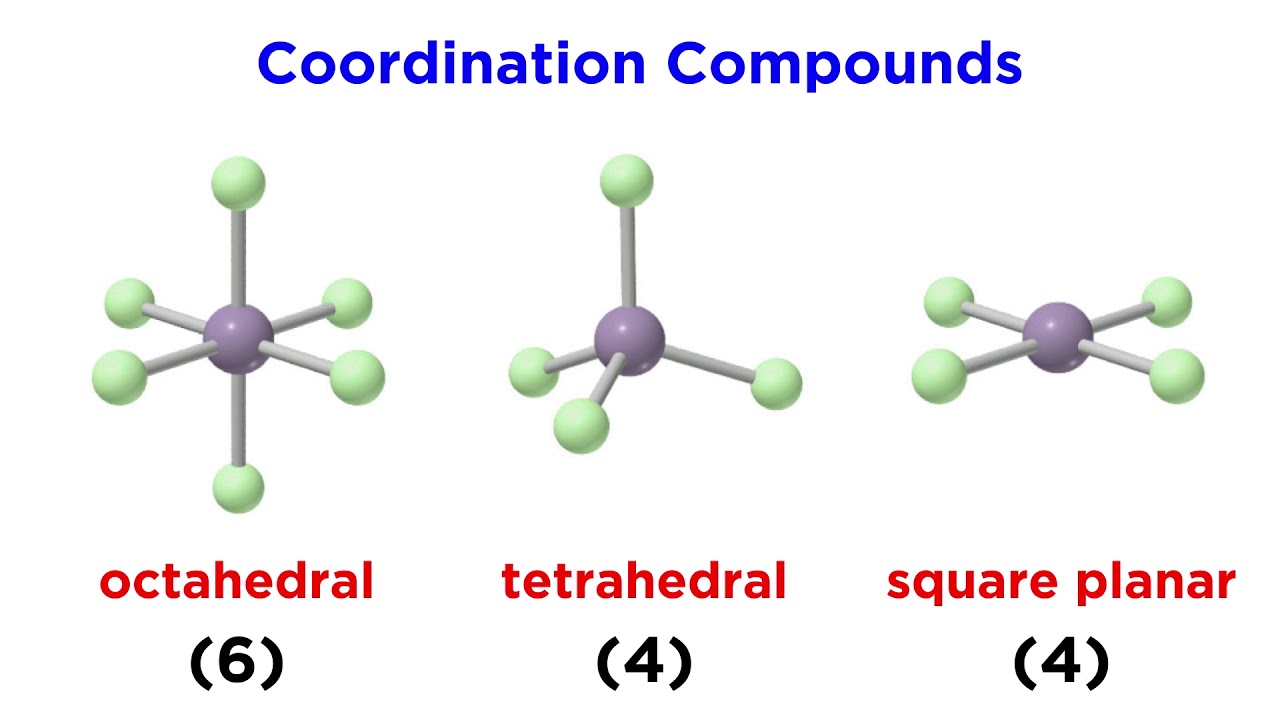
Coordination Compounds Geometry And Nomenclature One Young India Master the systematic naming of coordination compounds with step by step rules, tricks, and jee focused examples. The most common structures of the complexes in coordination compounds are square planar, tetrahedral, and octahedral, corresponding to coordination numbers of four, four, and six, respectively.
Comments are closed.