Converting Amines To Amides Chemistry Steps
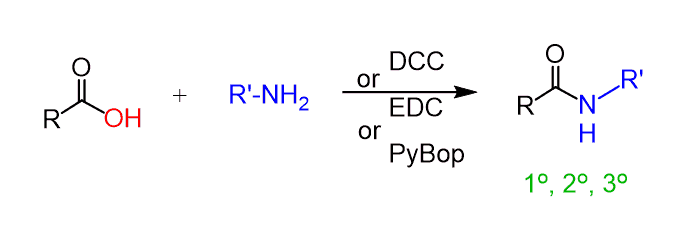
Converting Amines To Amides Chemistry Steps Check this post for more details and examples of reactions between acid chlorides and nucleophiles, as well as the linked articles for each of the methods mentioned for converting amines to amides. A list of common conditions for the conversion of an amine to an amide via acid chloride.

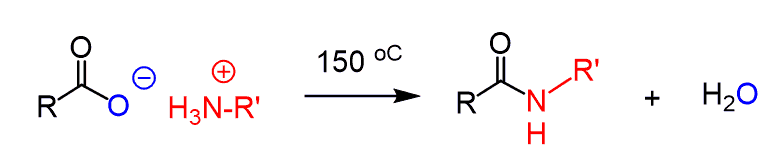
Converting Amines To Amides Chemistry Steps Simple amides can be prepared by reacting an acid anhydride with an amine. lastly, amides can be formed through the direct reaction of a carboxylic acid and an amine. however, this reaction is rarely used because the conditions are relatively severe. The carboxylic acid is first converted into an ammonium salt which then produces an amide on heating. the ammonium salt is formed by adding solid ammonium carbonate to an excess of the acid. Amides undergo hydrolysis to yield carboxylic acids plus ammonia or an amine upon heating in either aqueous acid or aqueous base. the conditions required for amide hydrolysis are more extreme than those required for the hydrolysis of acid chlorides or esters, but the mechanisms are similar. You can find more details about converting amides to aldehydes and ketones here. here is the summary for the preparation and most common reactions of amides, which you can use to work on the practice problems:.
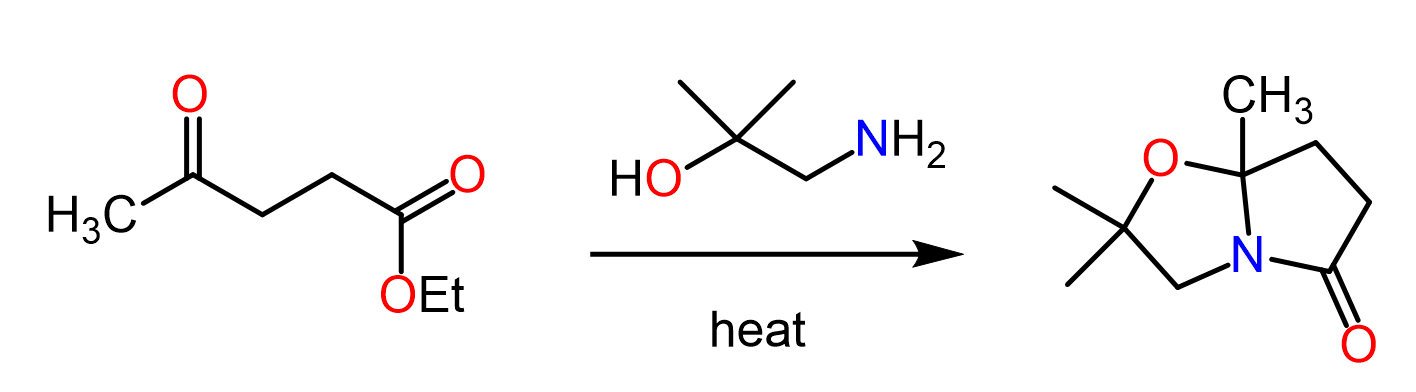
Converting Amines To Amides Chemistry Steps Amides undergo hydrolysis to yield carboxylic acids plus ammonia or an amine upon heating in either aqueous acid or aqueous base. the conditions required for amide hydrolysis are more extreme than those required for the hydrolysis of acid chlorides or esters, but the mechanisms are similar. You can find more details about converting amides to aldehydes and ketones here. here is the summary for the preparation and most common reactions of amides, which you can use to work on the practice problems:. In the following practice problems, the reactions and different ways of preparing amines will be discussed. keep in mind that aside from the reactions specifically related to amines, such as the gabriel amine synthesis, there are transformations that will require …. The first step is addition of nh 3 to the carbonyl (step 1, arrows a and b) to form a tetrahedral intermediate. this is followed by elimination of an alkoxide (step 2, arrows c and d) to give a protonated amide, which is then deprotonated (step 3, arrows e and f) to give the amide. A detailed mechanism illustrating the conversion of an amine to amide by first forming an acid chloride, then reacting with the amine. Amides undergo hydrolysis to yield carboxylic acids plus ammonia or an amine upon heating in either aqueous acid or aqueous base. the conditions required for amide hydrolysis are more extreme than those required for the hydrolysis of acid chlorides or esters, but the mechanisms are similar.
Converting Amines To Amides Chemistry Steps In the following practice problems, the reactions and different ways of preparing amines will be discussed. keep in mind that aside from the reactions specifically related to amines, such as the gabriel amine synthesis, there are transformations that will require …. The first step is addition of nh 3 to the carbonyl (step 1, arrows a and b) to form a tetrahedral intermediate. this is followed by elimination of an alkoxide (step 2, arrows c and d) to give a protonated amide, which is then deprotonated (step 3, arrows e and f) to give the amide. A detailed mechanism illustrating the conversion of an amine to amide by first forming an acid chloride, then reacting with the amine. Amides undergo hydrolysis to yield carboxylic acids plus ammonia or an amine upon heating in either aqueous acid or aqueous base. the conditions required for amide hydrolysis are more extreme than those required for the hydrolysis of acid chlorides or esters, but the mechanisms are similar.

Converting Amines To Amides Chemistry Steps A detailed mechanism illustrating the conversion of an amine to amide by first forming an acid chloride, then reacting with the amine. Amides undergo hydrolysis to yield carboxylic acids plus ammonia or an amine upon heating in either aqueous acid or aqueous base. the conditions required for amide hydrolysis are more extreme than those required for the hydrolysis of acid chlorides or esters, but the mechanisms are similar.
Comments are closed.