Colloids Definition Properties Classification Examples 54 Off

Colloids Definition Properties Classification Examples 54 Off What are colloids. learn its properties, types, and applications. check out a few examples of colloid fluids. Learn about the classification of colloids with clear types, examples, and properties explained for students.
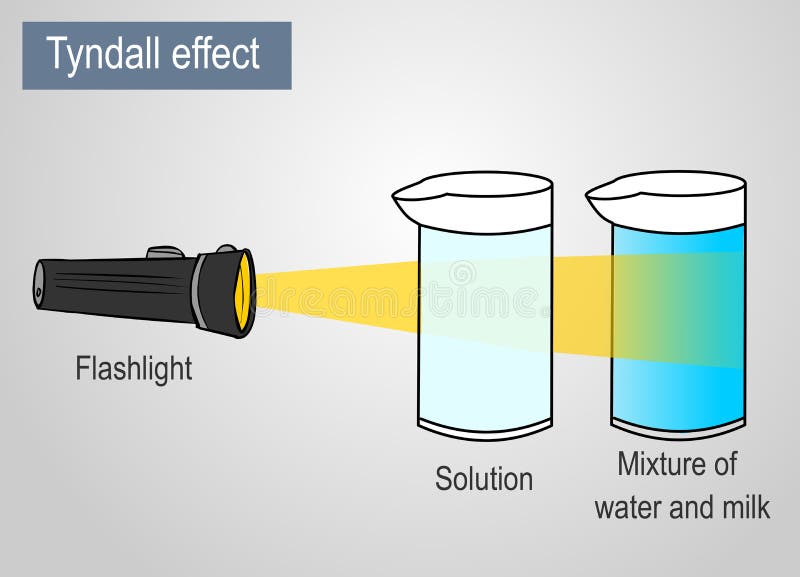
Colloids Definition Properties Classification Examples 54 Off In a colloid, the size of. Classification of colloids is done on the basis of three important factors: physical state, dispersion medium, interparticle interactions and properties of the sol particle. A colloid is one of the three primary types of mixtures, with the other two being a solution and suspension. a colloid is a mixture that has particles ranging between 1 and 1000 nanometers in diameter, yet are still able to remain evenly distributed throughout the solution. Learn what a colloid is in chemistry. get the definition and colloid examples. see how it differs from a solution or suspension.

Colloids Definition Properties Classification Examples 47 Off A colloid is one of the three primary types of mixtures, with the other two being a solution and suspension. a colloid is a mixture that has particles ranging between 1 and 1000 nanometers in diameter, yet are still able to remain evenly distributed throughout the solution. Learn what a colloid is in chemistry. get the definition and colloid examples. see how it differs from a solution or suspension. Classification of colloids explained with types, properties, and examples for easy understanding of dispersed systems. A colloid or colloidal solution is a type of mixture in which insoluble particles of one substance are uniformly dispersed in another substance. colloids play an important role in our daily life, although we often use them without realizing it. Classification of colloids the colloids are classified on the basis of the following criteria : 1) physical state of dispersed phase and dispersion medium. 2) natural of interactions between dispersed phase and dispersion medium. 3) type of particles of the dispersed phase. Based on their nature and composition, one can differentiate between three main classes of potentially hazardous colloidal particles: intrinsic colloidal particles, carrier colloidal particles, and biocolloids (grolimund et al. 2007).
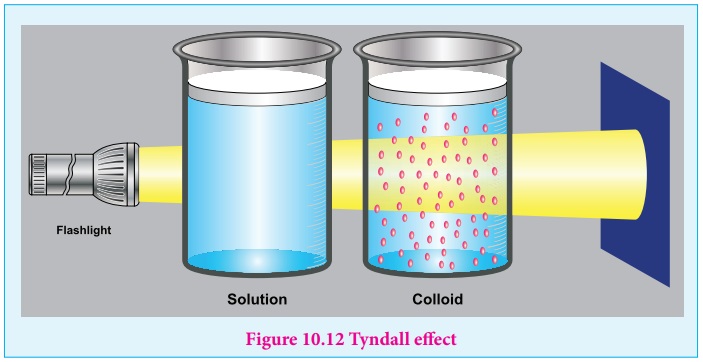
Colloids Definition Properties Classification Examples 47 Off Classification of colloids explained with types, properties, and examples for easy understanding of dispersed systems. A colloid or colloidal solution is a type of mixture in which insoluble particles of one substance are uniformly dispersed in another substance. colloids play an important role in our daily life, although we often use them without realizing it. Classification of colloids the colloids are classified on the basis of the following criteria : 1) physical state of dispersed phase and dispersion medium. 2) natural of interactions between dispersed phase and dispersion medium. 3) type of particles of the dispersed phase. Based on their nature and composition, one can differentiate between three main classes of potentially hazardous colloidal particles: intrinsic colloidal particles, carrier colloidal particles, and biocolloids (grolimund et al. 2007).

Colloids Definition Properties Classification Examples 54 Off Classification of colloids the colloids are classified on the basis of the following criteria : 1) physical state of dispersed phase and dispersion medium. 2) natural of interactions between dispersed phase and dispersion medium. 3) type of particles of the dispersed phase. Based on their nature and composition, one can differentiate between three main classes of potentially hazardous colloidal particles: intrinsic colloidal particles, carrier colloidal particles, and biocolloids (grolimund et al. 2007).
Comments are closed.