Colloids Definition Properties Classification Examples 47 Off

Colloids Definition Properties Classification Examples 54 Off What are colloids. learn its properties, types, and applications. check out a few examples of colloid fluids. Learn about the classification of colloids with clear types, examples, and properties explained for students.

Colloids Definition Properties Classification Examples 47 Off A colloid is one of the three primary types of mixtures, with the other two being a solution and suspension. a colloid is a mixture that has particles ranging between 1 and 1000 nanometers in diameter, yet are still able to remain evenly distributed throughout the solution. In a colloid, the size of. Learn what a colloid is in chemistry. get the definition and colloid examples. see how it differs from a solution or suspension. Colloids are mixtures where insoluble particles are suspended within another substance, with particle sizes ranging from 1 to 1000 nanometers. they can be classified based on physical states, interactions, properties, and dispersion mediums, with examples including emulsions, sols, and aerosols.
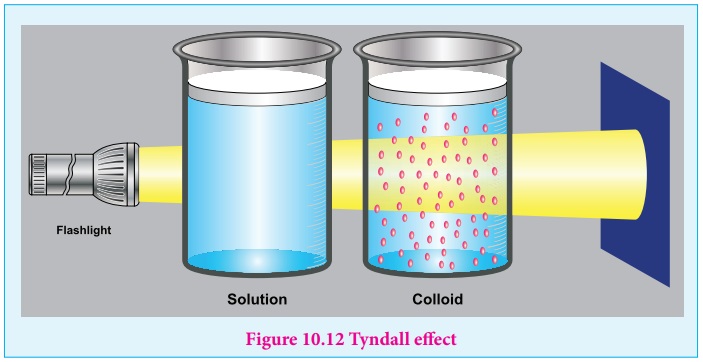
Colloids Definition Properties Classification Examples 47 Off Learn what a colloid is in chemistry. get the definition and colloid examples. see how it differs from a solution or suspension. Colloids are mixtures where insoluble particles are suspended within another substance, with particle sizes ranging from 1 to 1000 nanometers. they can be classified based on physical states, interactions, properties, and dispersion mediums, with examples including emulsions, sols, and aerosols. Suspension and colloid are both heterogeneous mixtures in which particles are dispersed in another substance; however, in a suspension the particles are large and settle down on standing, while in a colloid the particles are smaller and remain uniformly distributed without settling. Chemists classify these mixtures based on the size of their constituent particles, creating a spectrum from fully dissolved components to those that visibly separate. colloids represent a distinct, intermediate class within this spectrum, falling between true solutions and coarse suspensions. Colloids is a type of a mixture in which the particles of one substance (dispersed phase) are dispersed in another substance (dispersion medium). in this concept page, we will understand how colloids are different from other solutions, their classification, properties and applications. Colloids are unlike solutions because their dispersed particles are much larger than those of a solution. the dispersed particles of a colloid cannot be separated by filtration, but they scatter light, a phenomenon called the tyndall effect.
Colloids Definition Properties Classification Examples 47 Off Suspension and colloid are both heterogeneous mixtures in which particles are dispersed in another substance; however, in a suspension the particles are large and settle down on standing, while in a colloid the particles are smaller and remain uniformly distributed without settling. Chemists classify these mixtures based on the size of their constituent particles, creating a spectrum from fully dissolved components to those that visibly separate. colloids represent a distinct, intermediate class within this spectrum, falling between true solutions and coarse suspensions. Colloids is a type of a mixture in which the particles of one substance (dispersed phase) are dispersed in another substance (dispersion medium). in this concept page, we will understand how colloids are different from other solutions, their classification, properties and applications. Colloids are unlike solutions because their dispersed particles are much larger than those of a solution. the dispersed particles of a colloid cannot be separated by filtration, but they scatter light, a phenomenon called the tyndall effect.

Colloids Definition Properties Classification Examples 54 Off Colloids is a type of a mixture in which the particles of one substance (dispersed phase) are dispersed in another substance (dispersion medium). in this concept page, we will understand how colloids are different from other solutions, their classification, properties and applications. Colloids are unlike solutions because their dispersed particles are much larger than those of a solution. the dispersed particles of a colloid cannot be separated by filtration, but they scatter light, a phenomenon called the tyndall effect.
Comments are closed.