Clinical Trials Phases Explained A Complete Guide
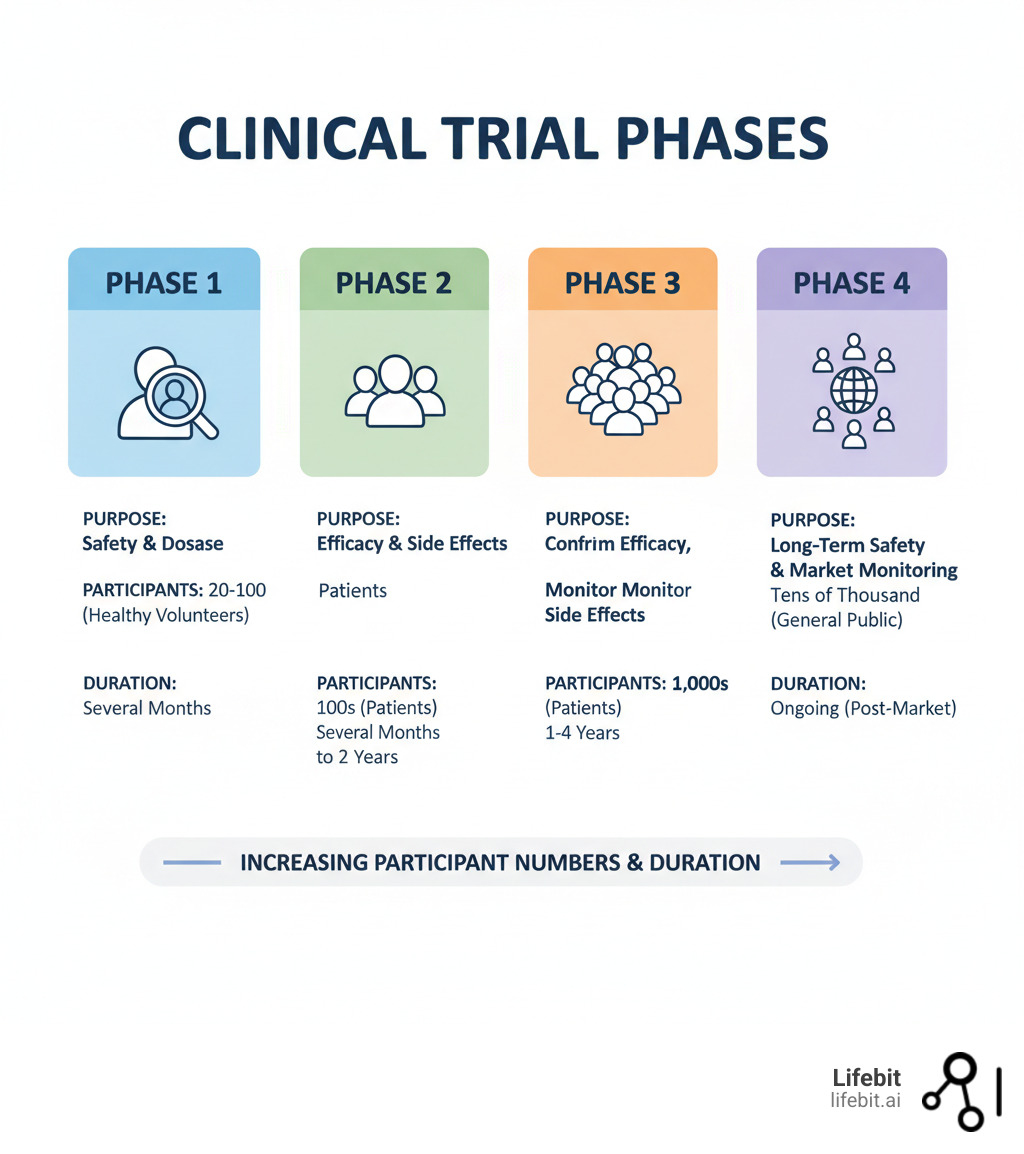
Clinical Trial 4 Phases Ultimate Guide There are 4 clinical trials phases, each with its specific goal. phase 1 ensures safety, while phase 2 studies effectiveness. phase 3 confirms large scale success, and phase 4 monitors real world safety. every phase minimizes risks before new treatments reach patients. An in depth guide to the four phases of clinical trials, updated for 2026. learn phase i iv objectives, fda single trial approval policy, ich e6 (r3) updates, ai driven trial design, and current success rates.

Clinical Trials Phases Explained A Complete Guide The 4 phases of clinical trials, from initial safety testing to post market monitoring. learn how each stage progresses, participant numbers, and objectives. Explore clinical trial phases from preclinical studies to post marketing surveillance, and understand their role in drug development and approval. Explore the four phases of clinical trials (phase i, ii, iii, iv), from initial human testing to post market surveillance. learn the purpose of each phase, participant numbers, and key objectives like safety, efficacy, and optimal dosing. What are the phases of clinical trials? clinical trials happen in four phases. researchers use each phase to determine if the treatment is safe and effective.

Phases Of Clinical Trials Explained Explore the four phases of clinical trials (phase i, ii, iii, iv), from initial human testing to post market surveillance. learn the purpose of each phase, participant numbers, and key objectives like safety, efficacy, and optimal dosing. What are the phases of clinical trials? clinical trials happen in four phases. researchers use each phase to determine if the treatment is safe and effective. A clear guide to clinical trial phases, from phase i to phase iv, explaining key goals and how each stage supports the overall drug development process. Learn about the different phases of clinical trials—from phase i to phase iv—and understand their purpose, processes, and importance with ccrps. Clinical trials are required before any new medicine can be approved for public use. this guide explains all clinical trial phases, from laboratory research to phase 4 monitoring, in simple terms. learn how safety, effectiveness, and approval are tested step by step. Researchers design clinical trials to answer specific research questions related to a medical product. these trials follow a specific study plan, called a protocol, that is developed by the.
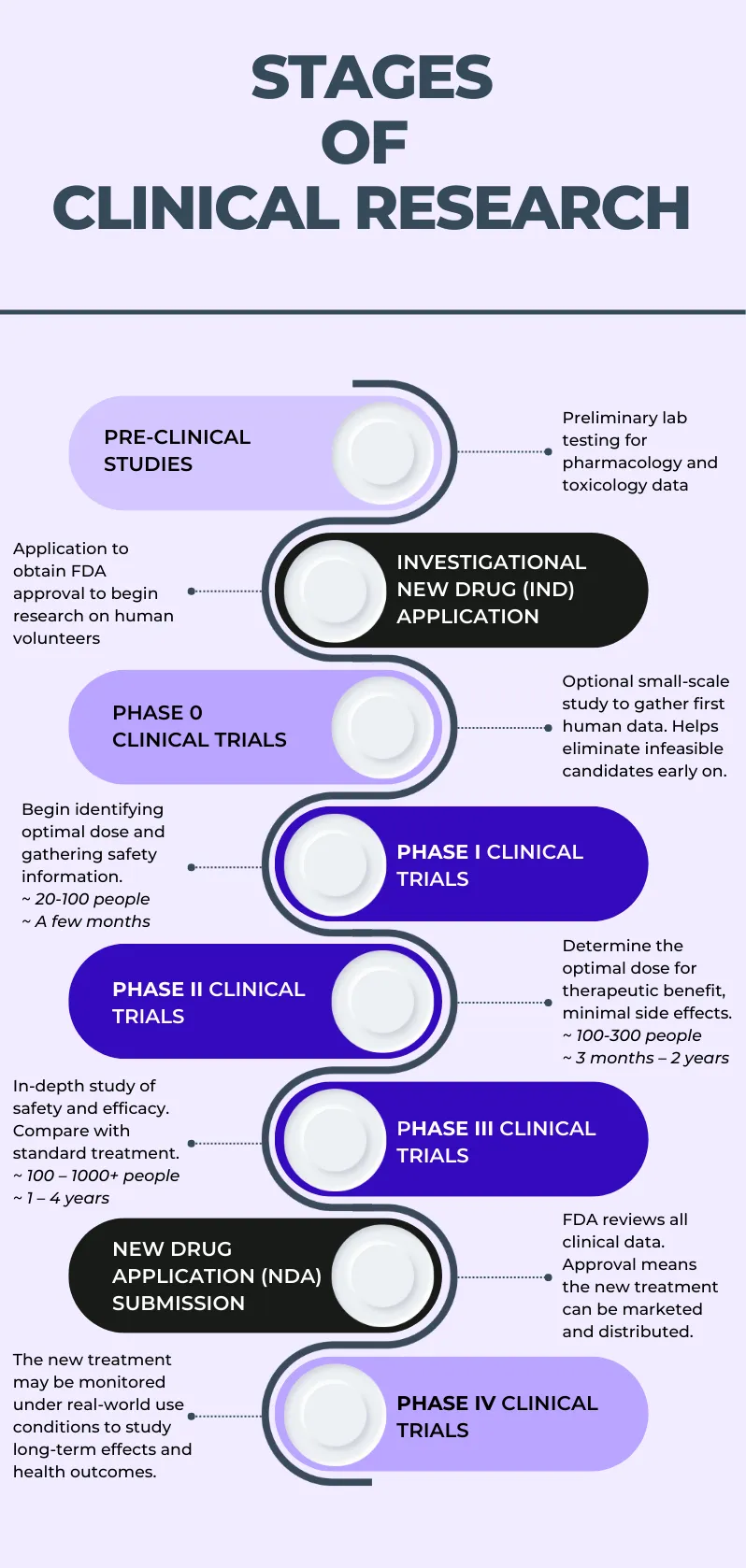
Clinical Trial Phases Explained The Ultimate Guide Power A clear guide to clinical trial phases, from phase i to phase iv, explaining key goals and how each stage supports the overall drug development process. Learn about the different phases of clinical trials—from phase i to phase iv—and understand their purpose, processes, and importance with ccrps. Clinical trials are required before any new medicine can be approved for public use. this guide explains all clinical trial phases, from laboratory research to phase 4 monitoring, in simple terms. learn how safety, effectiveness, and approval are tested step by step. Researchers design clinical trials to answer specific research questions related to a medical product. these trials follow a specific study plan, called a protocol, that is developed by the.
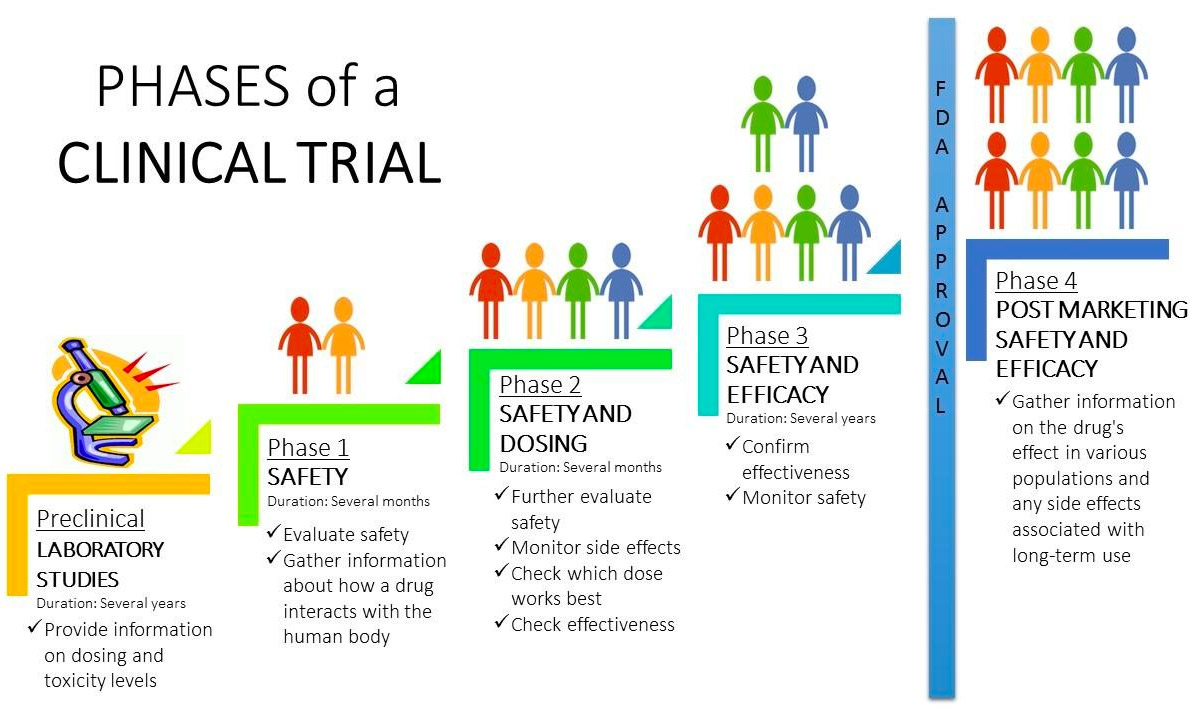
Cancer Clinical Trials Cancer Care St Luke S Cancer Center Clinical trials are required before any new medicine can be approved for public use. this guide explains all clinical trial phases, from laboratory research to phase 4 monitoring, in simple terms. learn how safety, effectiveness, and approval are tested step by step. Researchers design clinical trials to answer specific research questions related to a medical product. these trials follow a specific study plan, called a protocol, that is developed by the.
Comments are closed.