Clinical Trials Part 1

Lecture 4 Clinical Trials Part Ii Level 8 Pdf Clinical Trial Clinical trials are the scientific cornerstone of modern medicine, providing the evidence base for the safety and efficacy of new drugs and therapies. they are traditionally organized into four successive phases – i, ii, iii, and iv – each with distinct objectives, designs, and regulatory roles. Below, we explain the most common phases used in australian clinical trials, including investigator led or initiated trials. these phases apply to – but may differ between – trials for different interventions, from medical drugs and devices to diagnostic tools.

Clinical Trials Histologix Across all trial phases, the main expenses for clinical trials were administrative staff (about 20% of the total), clinical procedures (about 19%), and clinical monitoring of the subjects (about 11%). A phase of research to describe clinical trials that gather preliminary data on whether a drug works in people who have a certain condition disease (that is, the drug's effectiveness). The main aim of phase 1 trials is to find out about doses and side effects. they need to do this first, before testing the potential new treatment to see if it works. The first step in testing a new treatment is called a phase 1 clinical trial. researchers want to learn the right dosage of a drug that works best for a certain type of cancer.

Clinical Trials The main aim of phase 1 trials is to find out about doses and side effects. they need to do this first, before testing the potential new treatment to see if it works. The first step in testing a new treatment is called a phase 1 clinical trial. researchers want to learn the right dosage of a drug that works best for a certain type of cancer. Phase 1 trials study how an investigational treatment behaves in the body and what side effects it might cause. they start with low doses and slowly increase them, watching for harmful side effects. Phase 1 is the smallest and most controlled part of the process. it usually involves 20–100 healthy volunteers or patients. the main goal is to determine safety, dosage, and how the body processes the drug. here’s what typically happens in phase 1: volunteers receive very small doses at first. The 4 phases of clinical trials, from initial safety testing to post market monitoring. learn how each stage progresses, participant numbers, and objectives. This tutorial guides you through the essential components and best practices for designing a robust phase 1 clinical trial protocol. click to read the full article.
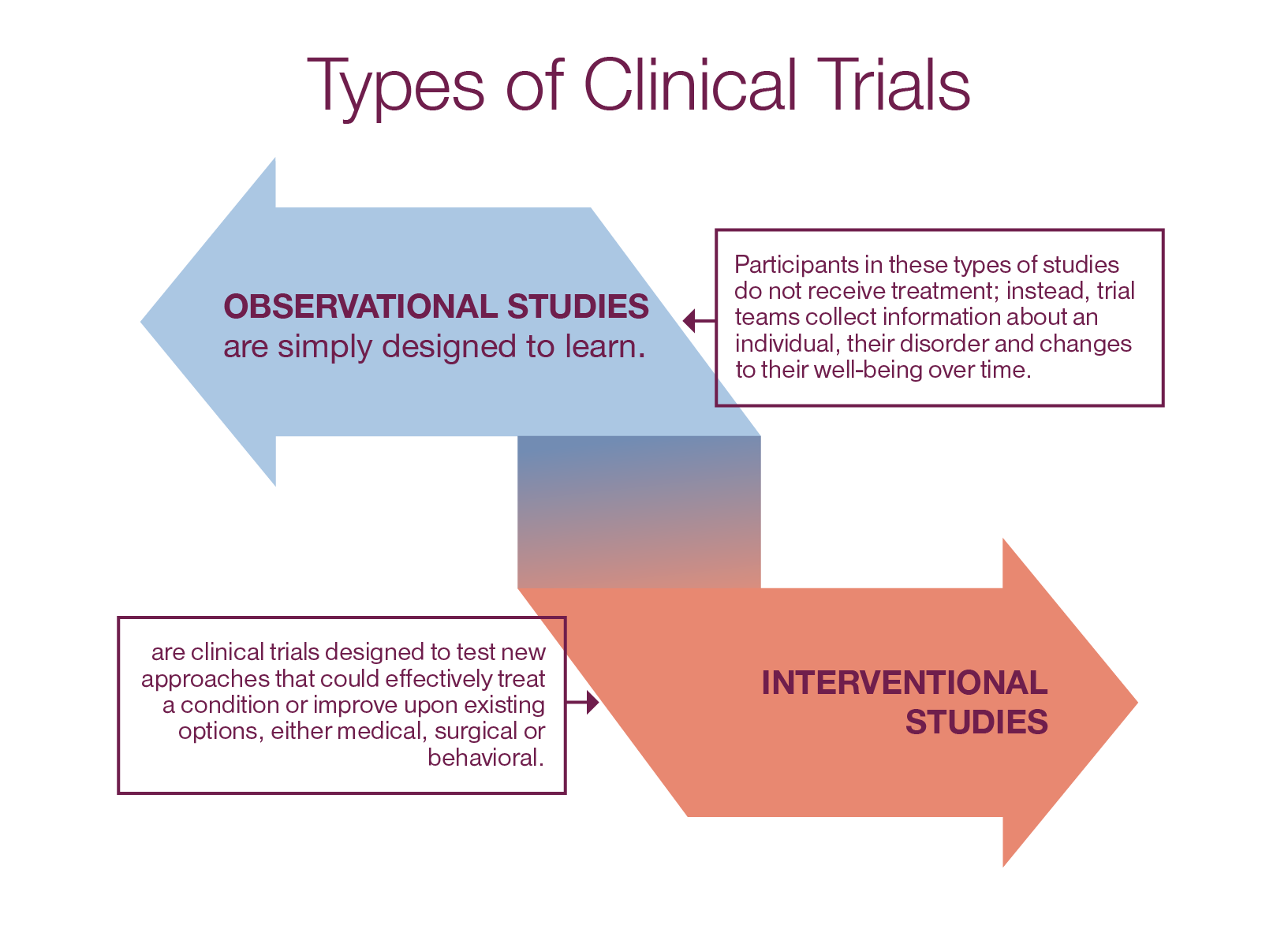
Clinical Trials Phase 1 trials study how an investigational treatment behaves in the body and what side effects it might cause. they start with low doses and slowly increase them, watching for harmful side effects. Phase 1 is the smallest and most controlled part of the process. it usually involves 20–100 healthy volunteers or patients. the main goal is to determine safety, dosage, and how the body processes the drug. here’s what typically happens in phase 1: volunteers receive very small doses at first. The 4 phases of clinical trials, from initial safety testing to post market monitoring. learn how each stage progresses, participant numbers, and objectives. This tutorial guides you through the essential components and best practices for designing a robust phase 1 clinical trial protocol. click to read the full article.
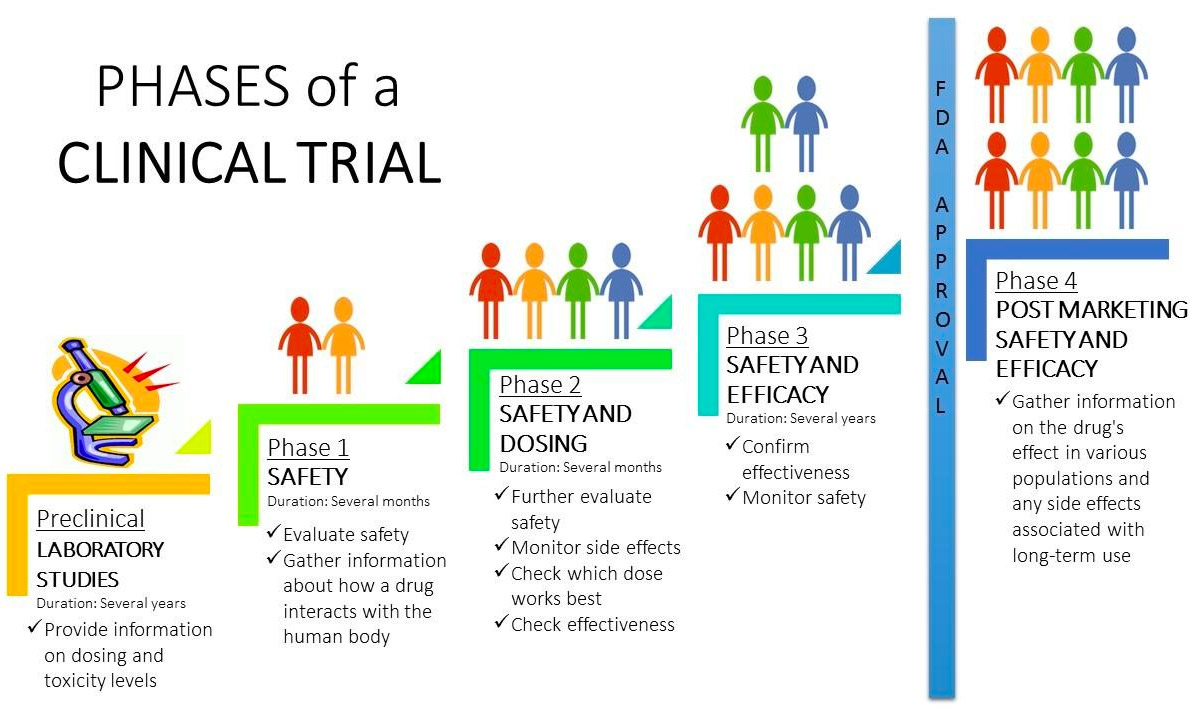
Clinical Trials The 4 phases of clinical trials, from initial safety testing to post market monitoring. learn how each stage progresses, participant numbers, and objectives. This tutorial guides you through the essential components and best practices for designing a robust phase 1 clinical trial protocol. click to read the full article.
Comments are closed.