Clinical Studies Tracker Presentation Layer

Clinical Tracker It is used for tracking the progress of trial subjects through multiple, independent, linear activities. the software was originally created by sheila raynor and kevin garwood and developed at the lifelong health and ageing unit of the medical research council. Use this ppt preset, which showcases a clinical study tracker, to provide a comprehensive overview of clinical trial activity, trends, and the status of trials within any healthcare system.
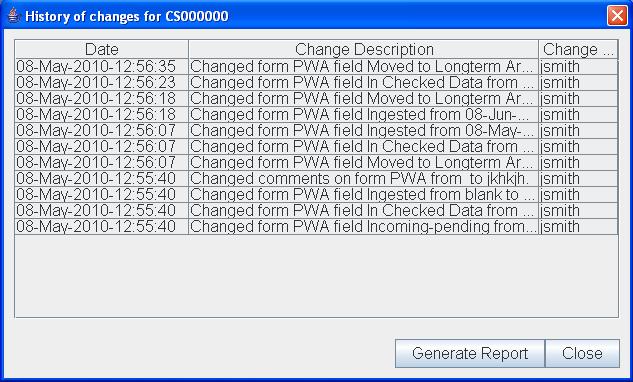
Clinical Studies Tracker Presentation Layer We reconfigure raw sas exports and complex tableau visualizations into mlr ready presentation slides, ensuring data fidelity is maintained while optimizing the visual hierarchy for immediate audience comprehension. Powergraph provides expert data modeling, custom power bi reporting, and specialized visuals designed specifically for the demands of clinical trials. clinical trials produce a vast array of data from diverse sources like edc, ctms, and labs. In this article, we explore how dashboards can be leveraged to monitor and visualize key performance indicators (kpis) across clinical trial sites. you’ll also learn best practices, key dashboard components, and how to ensure regulatory readiness with visual analytics tools. Access the code and data for the examples in this document here. deploying dashboards for monitoring clinical trials has many benefits. dashboards provide a simple way for investigators to keep track of study accrual and stratification factors.
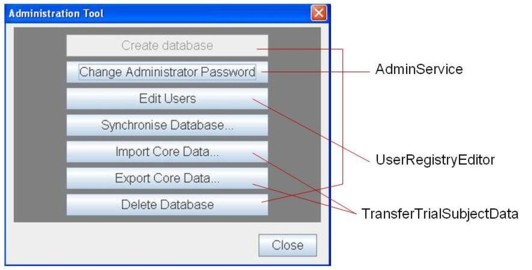
Clinical Studies Tracker Presentation Layer In this article, we explore how dashboards can be leveraged to monitor and visualize key performance indicators (kpis) across clinical trial sites. you’ll also learn best practices, key dashboard components, and how to ensure regulatory readiness with visual analytics tools. Access the code and data for the examples in this document here. deploying dashboards for monitoring clinical trials has many benefits. dashboards provide a simple way for investigators to keep track of study accrual and stratification factors. Interactive dashboards offer customizable views, enabling users to tailor the presentation of data to their specific requirements. this personalization enhances the user experience and ensures. Learn how to build a clinical data visualization dashboard for your clinical trial. better clinical data management in 5 steps. Medical monitors, safety review boards, and project teams can use these tools to effectively track and analyze key safety variables and study endpoints. Te fields to track during your clinical trial. can your system keep up with the demands of modern clinical trials? support 360° clinical trial management with a ctms that aggregates and reports on these fields faster and more eficiently than spreadsheet t.
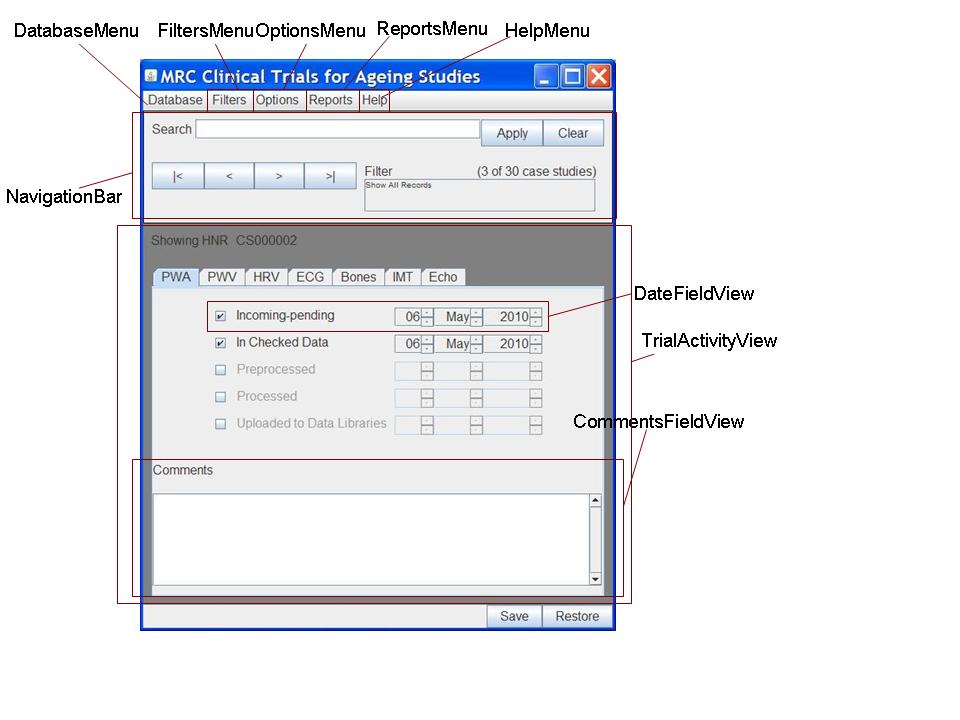
Clinical Studies Tracker Presentation Layer Interactive dashboards offer customizable views, enabling users to tailor the presentation of data to their specific requirements. this personalization enhances the user experience and ensures. Learn how to build a clinical data visualization dashboard for your clinical trial. better clinical data management in 5 steps. Medical monitors, safety review boards, and project teams can use these tools to effectively track and analyze key safety variables and study endpoints. Te fields to track during your clinical trial. can your system keep up with the demands of modern clinical trials? support 360° clinical trial management with a ctms that aggregates and reports on these fields faster and more eficiently than spreadsheet t.
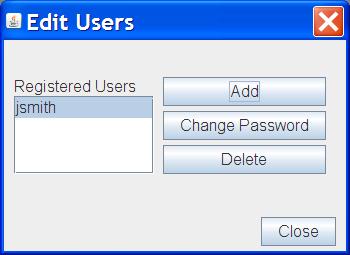
Clinical Studies Tracker Presentation Layer Medical monitors, safety review boards, and project teams can use these tools to effectively track and analyze key safety variables and study endpoints. Te fields to track during your clinical trial. can your system keep up with the demands of modern clinical trials? support 360° clinical trial management with a ctms that aggregates and reports on these fields faster and more eficiently than spreadsheet t.
Clinical Study Tracker Download Softpedia
Comments are closed.