Clinical Research Phase 1 Clinical Trials

Phase 1 Clinical Trials Moffitt Phase i clinical trials mark the critical first step in evaluating a new drug’s safety and tolerability in humans. these trials set the groundwork for all subsequent phases, determining whether a compound has the potential to advance toward broader patient populations. Under the predominant model, clinical development of a new drug proceeds through four phases: phase i: first in human trials (fih) for safety and dose finding. small cohorts (often 20–100 subjects) are given single or escalating doses.
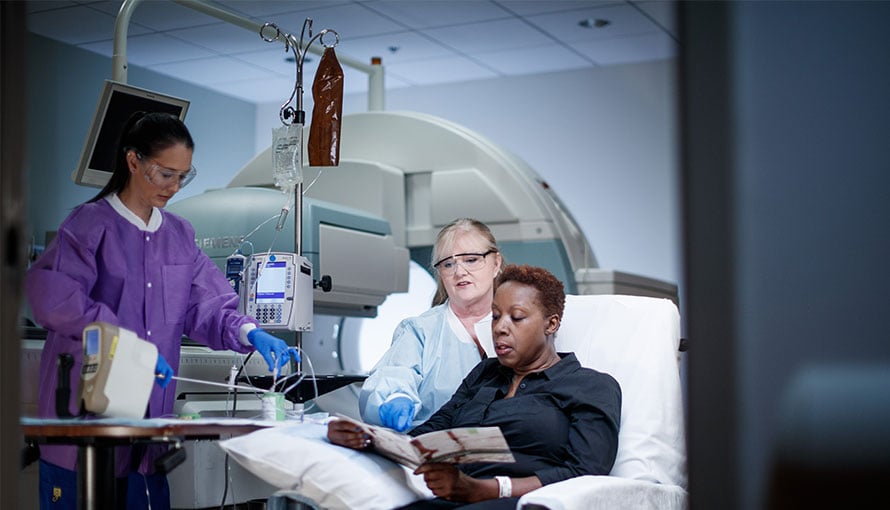
Phase 1 Clinical Trials Moffitt A phase of research to describe clinical trials that focus on the safety of a drug. they are usually conducted with healthy volunteers, and the goal is to determine the drug's most frequent and serious adverse events and, often, how the drug is broken down and excreted by the body. Across all trial phases, the main expenses for clinical trials were administrative staff (about 20% of the total), clinical procedures (about 19%), and clinical monitoring of the subjects (about 11%). The main aim of phase 1 trials is to find out about doses and side effects. they need to do this first, before testing the potential new treatment to see if it works. There are usually four phases of trials in humans (clinical trials). a phase 1 trial is usually the first time that an imp is tested in humans and so it will usually investigate the safe dose range and potential side effects, how it is metabolised and whether it might work in patients.

Phase 1 Clinical Trials Clinical Trials Bulgaria The main aim of phase 1 trials is to find out about doses and side effects. they need to do this first, before testing the potential new treatment to see if it works. There are usually four phases of trials in humans (clinical trials). a phase 1 trial is usually the first time that an imp is tested in humans and so it will usually investigate the safe dose range and potential side effects, how it is metabolised and whether it might work in patients. A phase 1 clinical trial is the first time an experimental drug or treatment is tested in humans. its primary goal is safety: researchers want to find out whether a new compound is safe enough to keep testing, what side effects it causes, and how the human body processes it. In phase 1 trials, the pool of testing is limited to a very small number of trial participants—healthy participants or patients. this phase of clinical trials focuses on “dose ranging”, wherein the ideal doses that would be safe for the end users or patients can be determined. A phase 1 clinical trial is the first time an investigational drug is tested in humans. it follows years of preclinical research — lab studies and animal testing — and represents the transition from theory to human biology. Clinical trials are focused on the observation and treatment of living patients, rather than laboratory or theoretical research. however, many clinical trials have an early phase that may involve clinical or non clinical research, from small human studies to animal testing.

Understanding Phase I Clinical Trials A Guide For Patients A phase 1 clinical trial is the first time an experimental drug or treatment is tested in humans. its primary goal is safety: researchers want to find out whether a new compound is safe enough to keep testing, what side effects it causes, and how the human body processes it. In phase 1 trials, the pool of testing is limited to a very small number of trial participants—healthy participants or patients. this phase of clinical trials focuses on “dose ranging”, wherein the ideal doses that would be safe for the end users or patients can be determined. A phase 1 clinical trial is the first time an investigational drug is tested in humans. it follows years of preclinical research — lab studies and animal testing — and represents the transition from theory to human biology. Clinical trials are focused on the observation and treatment of living patients, rather than laboratory or theoretical research. however, many clinical trials have an early phase that may involve clinical or non clinical research, from small human studies to animal testing.

Phase1 Clinical Trials Clinical Trials Bulgaria A phase 1 clinical trial is the first time an investigational drug is tested in humans. it follows years of preclinical research — lab studies and animal testing — and represents the transition from theory to human biology. Clinical trials are focused on the observation and treatment of living patients, rather than laboratory or theoretical research. however, many clinical trials have an early phase that may involve clinical or non clinical research, from small human studies to animal testing.

Phase 1 Clinical Trials Files Sofpromed
Comments are closed.