Class I Mdr

Class 1 Medical Device Requirements Oriel Stat A Matrix Viii of regulation (eu) 2017 745 on medical devices (mdr). they correspond, to a large extent, to the classification rules established by the international medical device regulators. The european union medical device regulation (eu mdr) categorizes medical devices into one of four classes: class i, class iia, class iib, and class iii medical devices. the mdr medical device classification is based on the device’s potential risk of harm to users.

Class 1 Medical Device Requirements Oriel Stat A Matrix The complete walkthrough: mdr class i example devices, where to found your company (not in berlin!), how much it costs and how long it takes. This is an overview of relevant topics for all manufacturers of class 1 medical devices under eu mdr 2017 745 in alignment with mdcg 2019 15. Class i medical devices pose almost no risk to human safety. these are further sub divided into four sub classes: class i medical devices include non sterile devices and medical devices without a measurement function (e.g., facial mask, gauze, gel for ultrasound). Explore eu mdr class i medical device requirements, including self certification, qms, pms, udi, and best practices for low risk product compliance.
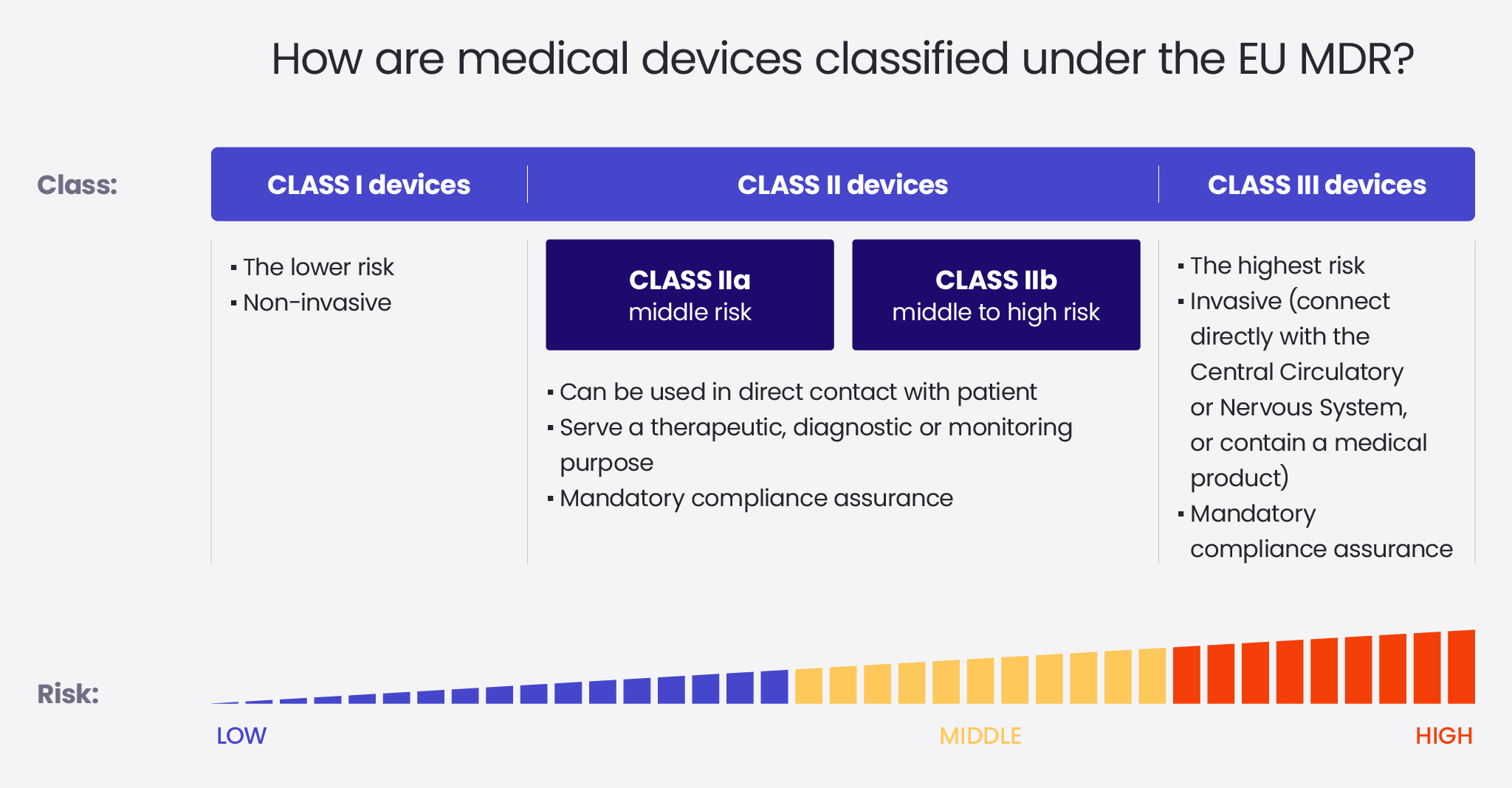
The Complete Guide To Eu Medical Device Regulation Spyrosoft Class i medical devices pose almost no risk to human safety. these are further sub divided into four sub classes: class i medical devices include non sterile devices and medical devices without a measurement function (e.g., facial mask, gauze, gel for ultrasound). Explore eu mdr class i medical device requirements, including self certification, qms, pms, udi, and best practices for low risk product compliance. Class i (non sterile, non measuring) low risk devices that do not require notified body involvement for ce marking. examples include bandages and stethoscopes. Class i: this group includes low risk devices like non invasive devices, such as hospital beds or bandages, or invasive tools used only transiently in body orifices, such as dental impression materials. class i devices can be active, such as examination lamps. In medical device classification eu mdr, devices are segmented into four classes: class i, class iia, class iib, and class iii. they span from low risk items such as reusable surgical instruments (class i) through high risk implants such as pacemakers (class iii). Class i (low risk): the devices in this category bear very minimal risk to both users and patients. most class i devices, except those that are sterile, have a measuring function, or are reusable surgical instruments do not require notified body intervention for any type of support.

The Complete Guide To Eu Medical Device Regulation Spyrosoft Class i (non sterile, non measuring) low risk devices that do not require notified body involvement for ce marking. examples include bandages and stethoscopes. Class i: this group includes low risk devices like non invasive devices, such as hospital beds or bandages, or invasive tools used only transiently in body orifices, such as dental impression materials. class i devices can be active, such as examination lamps. In medical device classification eu mdr, devices are segmented into four classes: class i, class iia, class iib, and class iii. they span from low risk items such as reusable surgical instruments (class i) through high risk implants such as pacemakers (class iii). Class i (low risk): the devices in this category bear very minimal risk to both users and patients. most class i devices, except those that are sterile, have a measuring function, or are reusable surgical instruments do not require notified body intervention for any type of support.
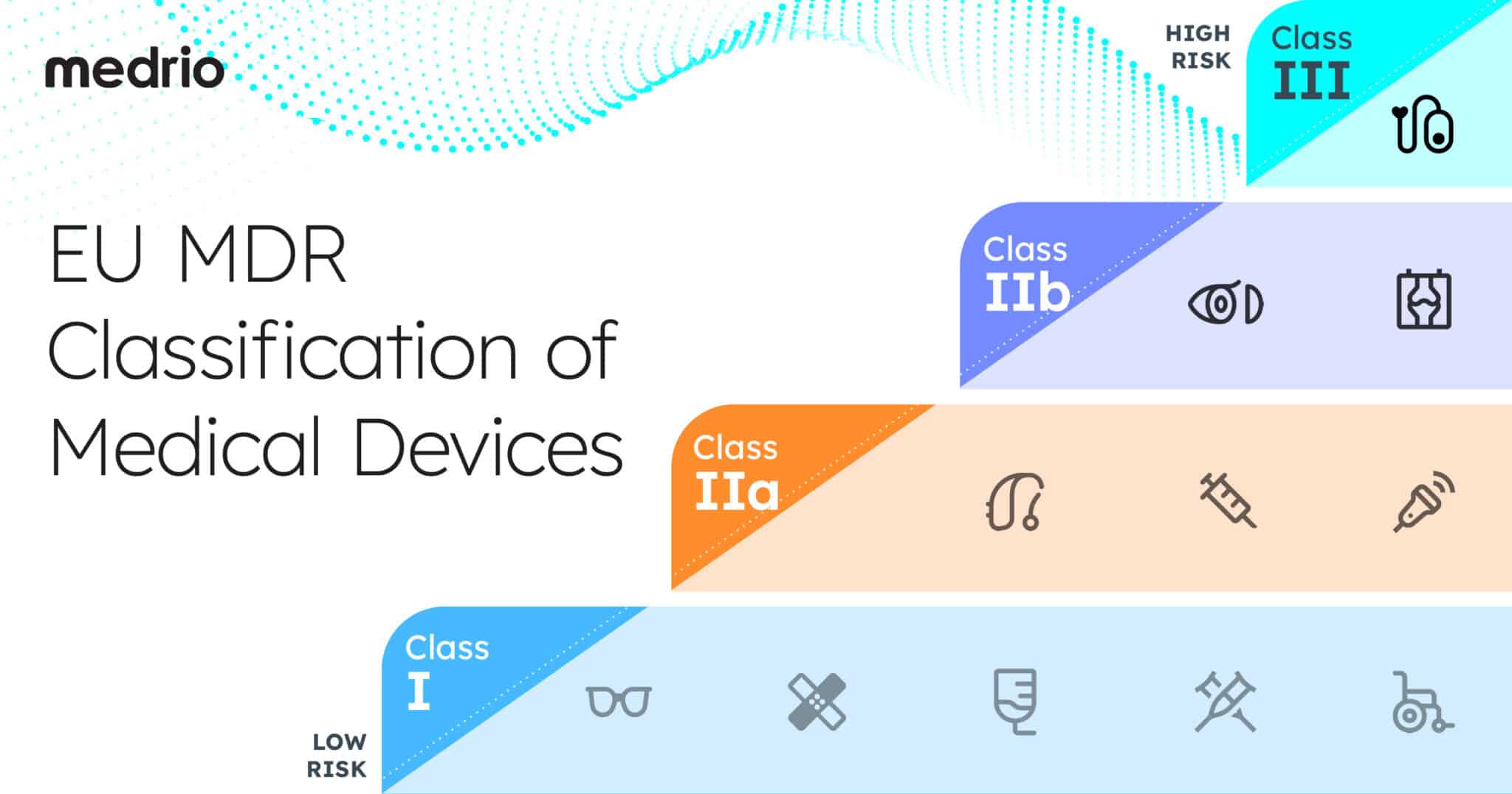
European Mdr Eu Mdr Compliance Guide In medical device classification eu mdr, devices are segmented into four classes: class i, class iia, class iib, and class iii. they span from low risk items such as reusable surgical instruments (class i) through high risk implants such as pacemakers (class iii). Class i (low risk): the devices in this category bear very minimal risk to both users and patients. most class i devices, except those that are sterile, have a measuring function, or are reusable surgical instruments do not require notified body intervention for any type of support.
Comments are closed.