Chemical Properties Of Matter Definition And Examples

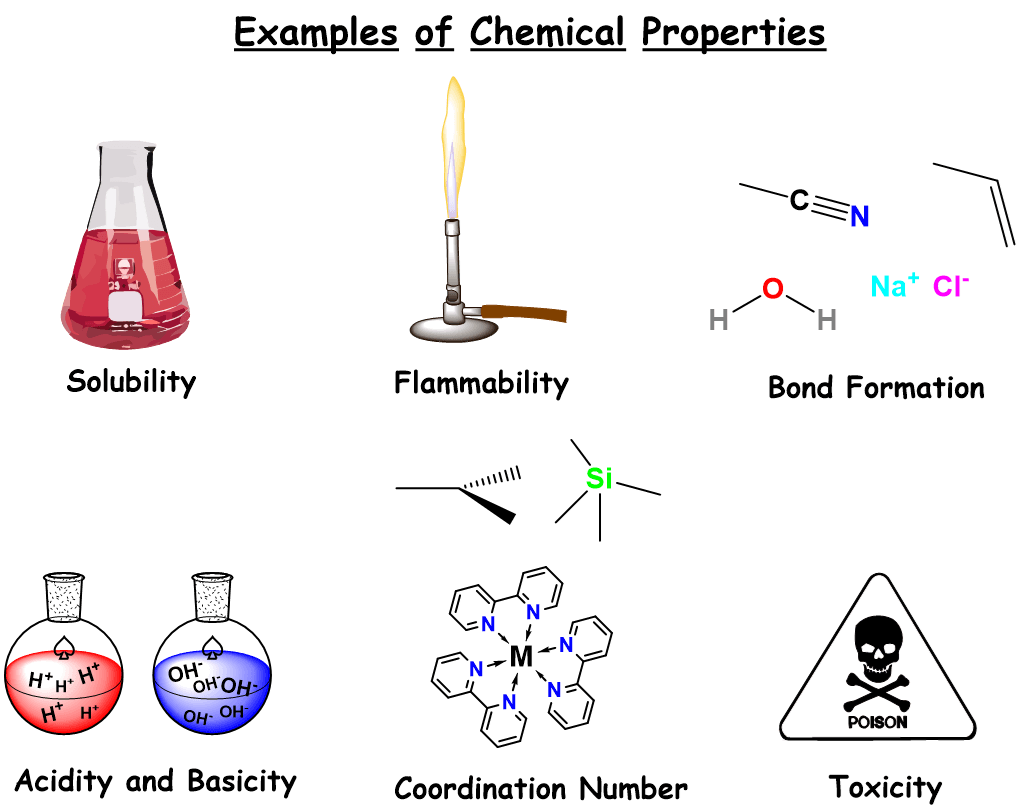
Chemical Properties Of Matter Definition And Examples Chemical By studying chemical properties, scientists can classify compounds and predict their behavior, reactions, and uses. they can separate a substance from other substances and purify it. Key points: chemical property a chemical property describes how a substance behaves in a chemical reaction or transforms into a new substance. these properties are only observable through a chemical change. examples include flammability, reactivity, and oxidation state.
Physical Vs Chemical Properties Definition Examples Expii Understanding the chemical properties of matter via explanations and examples can reveal how substances behave and transform in chemical reactions. Learn about chemical properties of matter with simple explanations, examples, and interactive quizzes. perfect for students studying chemistry concepts. This lesson discusses the distinction between physical properties like density, melting and boiling points, color, and conductivity and chemical properties which describe how substances react or change. Chemical properties of matter describes its "potential" to undergo some chemical change or reaction by virtue of its composition. what elements, electrons, and bonding are present to give the potential for chemical change.
/chemical-properties-of-matter-608337-v33-5b6334d346e0fb0082054666.png)
Chemical Properties Of Matter This lesson discusses the distinction between physical properties like density, melting and boiling points, color, and conductivity and chemical properties which describe how substances react or change. Chemical properties of matter describes its "potential" to undergo some chemical change or reaction by virtue of its composition. what elements, electrons, and bonding are present to give the potential for chemical change. Chemical properties are properties that can be measured or observed only when matter undergoes a change to become an entirely different kind of matter. for example, the ability of iron to rust can only be observed when iron actually rusts. The ability of a substance to undergo a chemical change is determined by the arrangement and behavior of its atoms and molecules, which dictates the type of bonds it can form or break. chemical properties govern how a material will interact with its environment to form new substances. In this tutorial, you have known about properties of matter: physical and chemical with various examples. hope that you will have understood the basic points of intensive and extensive characteristics. The change of one type of matter into another type (or the inability to change) is a chemical property. examples of chemical properties include flammability, toxicity, acidity, reactivity (many types), and heat of combustion.
Comments are closed.