Chemical Equilibrium
Chemical Equilibrium Definition Types Importance And 50 Off When a chemical reaction is at equilibrium, any disturbance of the system, such as a change in temperature, or addition or removal of one of the reaction components, will "shift" the composition to a new equilibrium state. Learn chemical equilibrium with equations, examples, diagrams, and applications. also, learn the law of chemical equilibrium and the chemical constant.

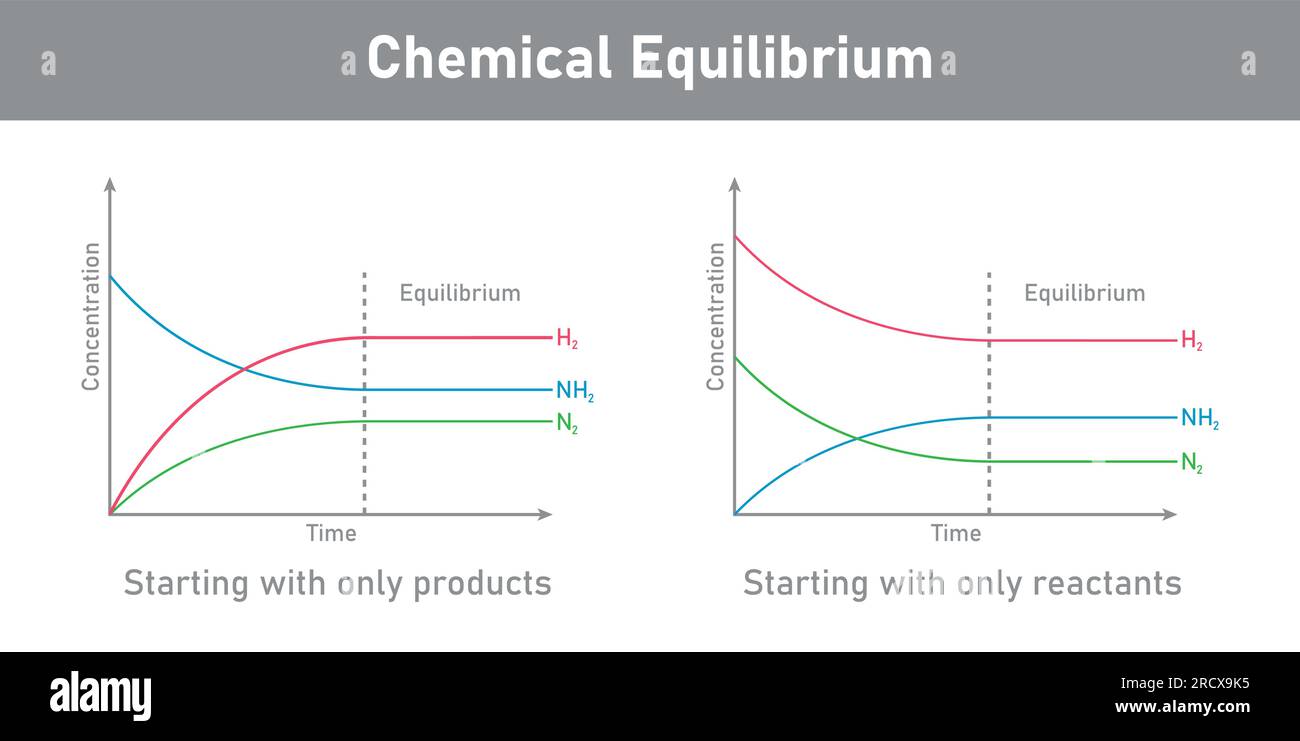
Chemical Equilibrium Quiz Quiz Now Learn about the concept, history, and applications of chemical equilibrium, the state in which reactants and products are present in constant concentrations. find out how equilibrium is related to reaction rates, gibbs free energy, and le châtelier's principle. Chemical equilibrium occurs when a reaction and its reverse reaction proceed at the same rate. in the figure above, equilibrium is finally reached in the third picture. as a system approaches equilibrium, both the forward and reverse reactions are occurring. Chemical equilibrium is the condition in the course of a reversible chemical reaction in which no net change in the amounts of reactants and products occurs. a reversible chemical reaction is one in which the products, as soon as they are formed, react to produce the original reactants. Learn what equilibrium is in chemistry, how it works, and why it matters. explore different types of equilibrium, such as physical, solubility, and acid base, and how they affect reactions in industry and biology.
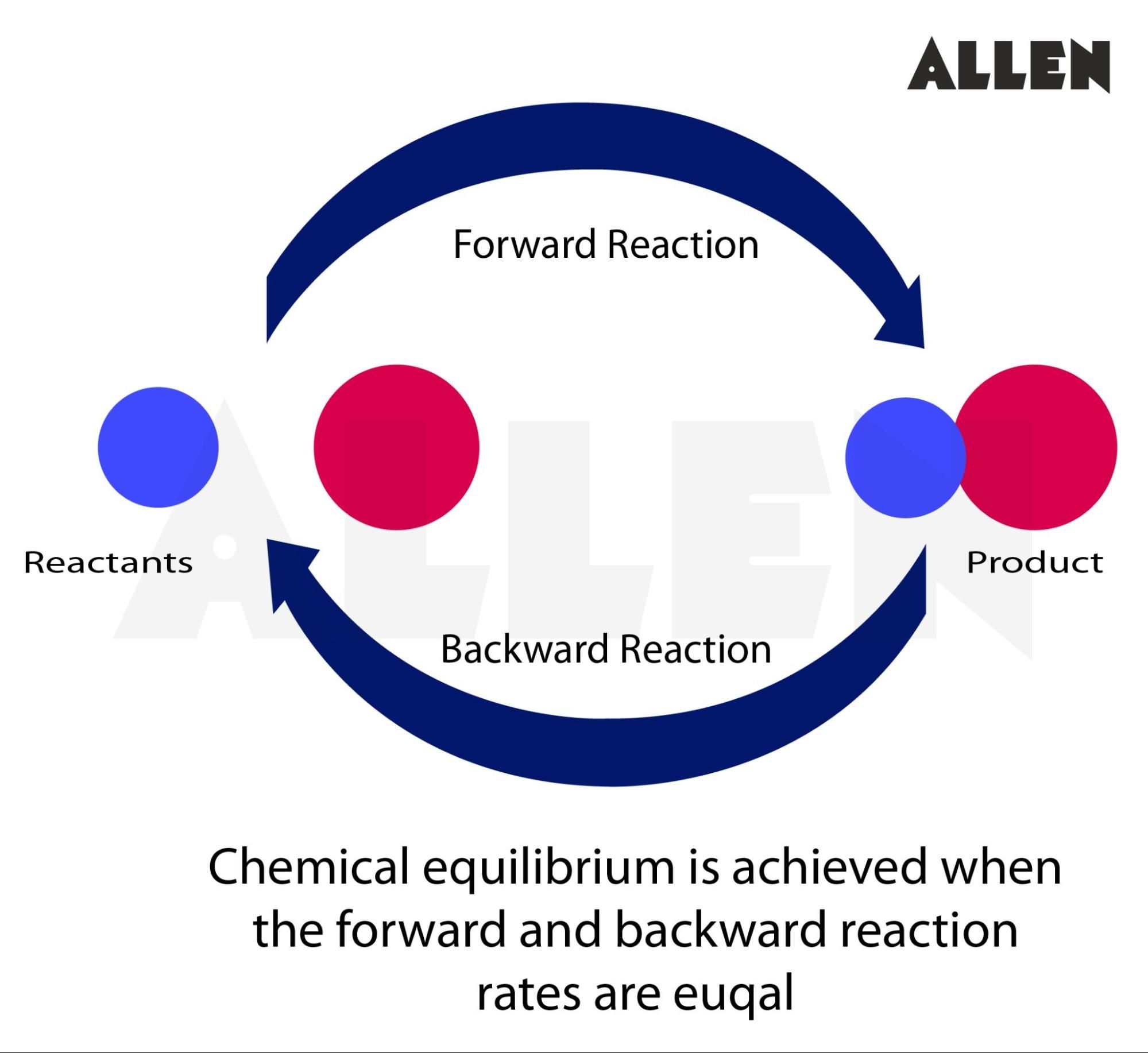
Chemical Equilibrium Importance Types And Faqs Chemical equilibrium is the condition in the course of a reversible chemical reaction in which no net change in the amounts of reactants and products occurs. a reversible chemical reaction is one in which the products, as soon as they are formed, react to produce the original reactants. Learn what equilibrium is in chemistry, how it works, and why it matters. explore different types of equilibrium, such as physical, solubility, and acid base, and how they affect reactions in industry and biology. Learn the concept, equilibrium constant, and expressions of chemical equilibrium, as well as the effect of changes in concentration, pressure, volume, and temperature. explore the dynamic and reversible nature of equilibrium and the le chatelier's principle. Chemical equilibrium refers to a dynamic state in which the forward and reverse reaction rates are equal, so concentrations of reactants and products remain constant — though there is still change occurring at the molecular level. Learn what chemical equilibrium is, how it works, and why it matters with clear examples and key concepts for students. Any reversible chemical reaction can achieve equilibrium, which for such a reaction is a dynamic state. it is best for us to approach an understanding of equilibrium reactions by looking at some experiments—this is how the early chemists did it.

Presentation Chemical Equilibrium Cover Stable Diffusion Online Learn the concept, equilibrium constant, and expressions of chemical equilibrium, as well as the effect of changes in concentration, pressure, volume, and temperature. explore the dynamic and reversible nature of equilibrium and the le chatelier's principle. Chemical equilibrium refers to a dynamic state in which the forward and reverse reaction rates are equal, so concentrations of reactants and products remain constant — though there is still change occurring at the molecular level. Learn what chemical equilibrium is, how it works, and why it matters with clear examples and key concepts for students. Any reversible chemical reaction can achieve equilibrium, which for such a reaction is a dynamic state. it is best for us to approach an understanding of equilibrium reactions by looking at some experiments—this is how the early chemists did it.

Honors Chem Chemical Equilibrium Flashcards Quizlet Learn what chemical equilibrium is, how it works, and why it matters with clear examples and key concepts for students. Any reversible chemical reaction can achieve equilibrium, which for such a reaction is a dynamic state. it is best for us to approach an understanding of equilibrium reactions by looking at some experiments—this is how the early chemists did it.
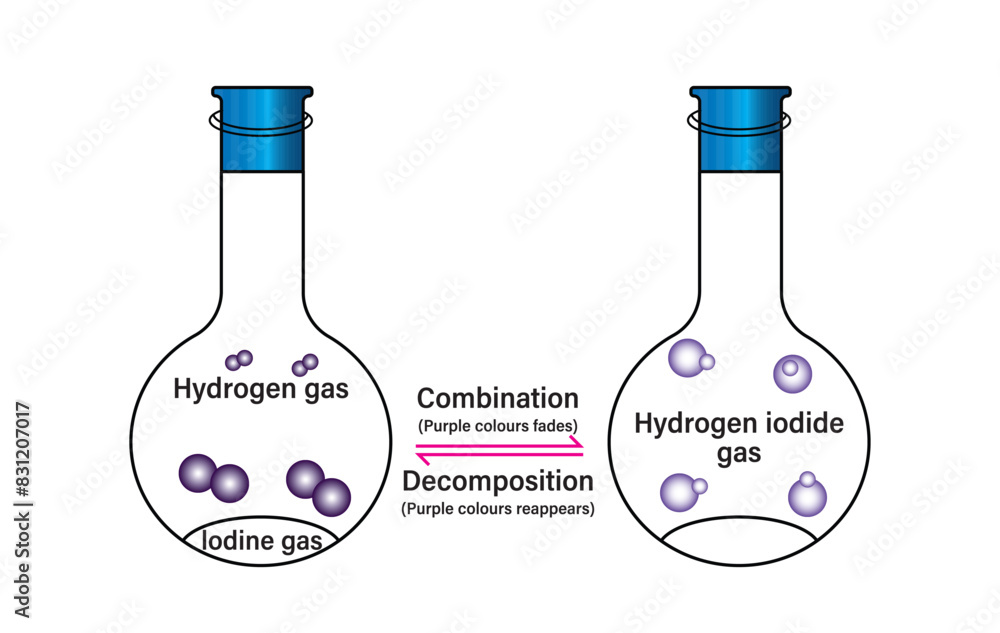
Reversible Reactions Reversible Reaction And Dynamic Equilibrium
Comments are closed.