Checkmate 568 Efficacy And Biomarker Analysis For Nivolumab And
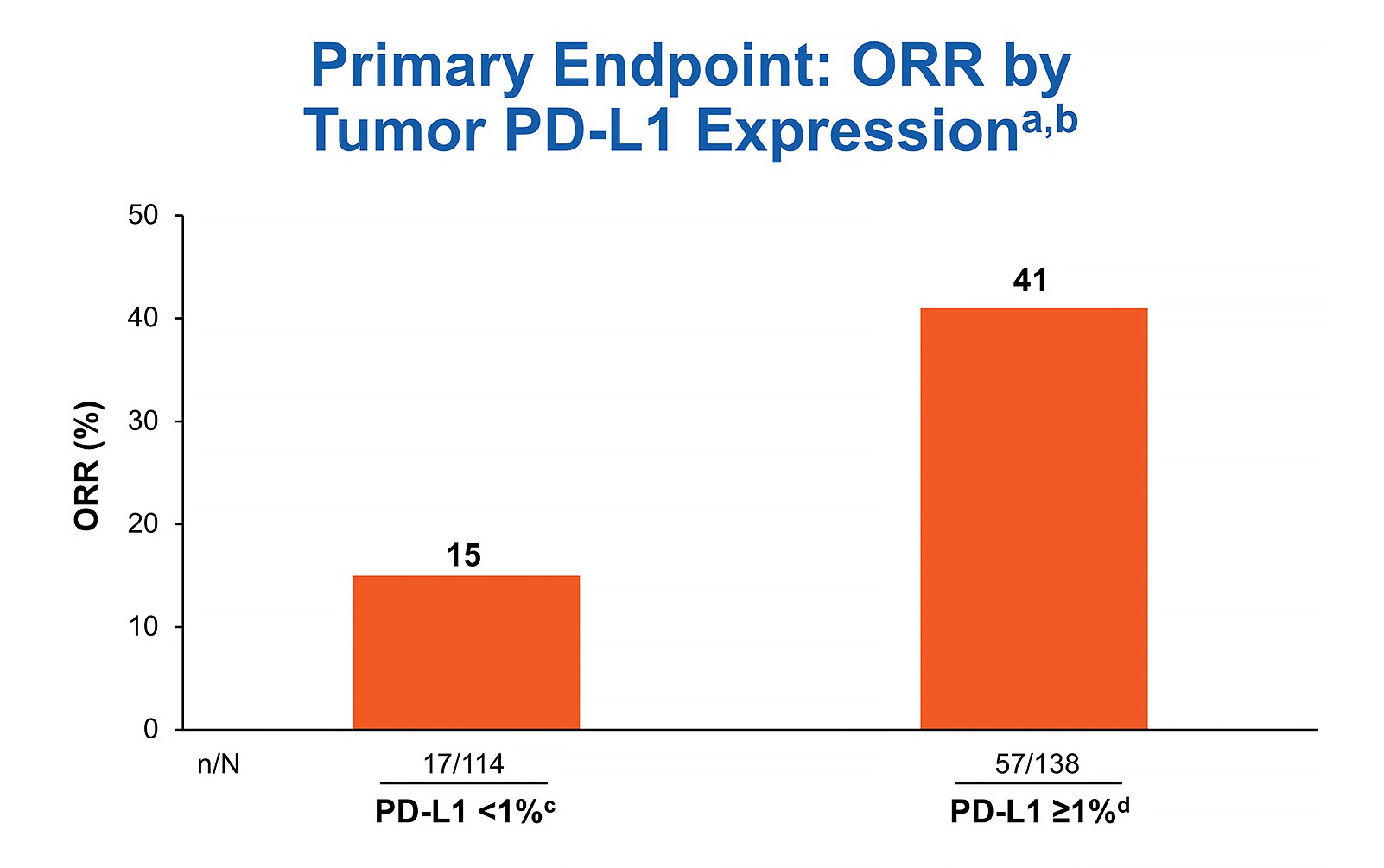
Checkmate 568 Efficacy And Biomarker Analysis For Nivolumab And Checkmate 568 demonstrates that the combination of nivolumab 3 mg kg every 2 weeks plus ipilimumab 1 mg kg every 6 weeks was tolerable with superior efficacy compared to historical outcomes for nivolumab monotherapy in untreated advanced stage nsclc. Checkmate 568 is an open label phase ii trial that evaluated the efficacy and safety of nivolumab plus low dose ipilimumab as first line treatment of advanced metastatic non–small cell lung cancer (nsclc).
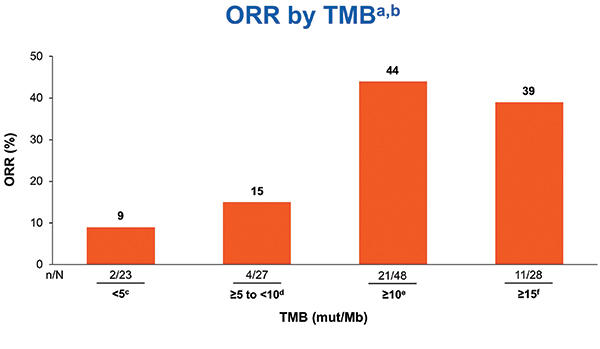
Checkmate 568 Efficacy And Biomarker Analysis For Nivolumab And Purpose checkmate 568 is an open label phase ii trial that evaluated the efficacy and safety of nivolumab plus low dose ipilimumab as rst line treatment of advanced metastatic non–small cell lung cancer (nsclc). We characterized the safety of first line nivolumab plus ipilimumab (nivo ipi) in a large patient population with metastatic nsclc and efficacy outcomes after nivo ipi discontinuation owing to treatment related adverse events (traes). Nivolumab plus ipilimumab or nivolumab alone versus ipilimumab alone in advanced melanoma (checkmate 067): 4 year outcomes of a multicentre, randomised, phase 3 trial. Purpose checkmate 568 is an open label phase ii trial that evaluated the efficacy and safety of nivolumab plus low dose ipilimumab as first line treatment of advanced metastatic non small cell lung cancer (nsclc).

Checkmate 568 Efficacy And Biomarker Analysis For Nivolumab And Nivolumab plus ipilimumab or nivolumab alone versus ipilimumab alone in advanced melanoma (checkmate 067): 4 year outcomes of a multicentre, randomised, phase 3 trial. Purpose checkmate 568 is an open label phase ii trial that evaluated the efficacy and safety of nivolumab plus low dose ipilimumab as first line treatment of advanced metastatic non small cell lung cancer (nsclc). Here, we report the efficacy of first line nivolumab plus ipilimumab treatment in a large, pooled population of patients with advanced nsclc from checkmate 227 part 1, checkmate 817 (cohort a), checkmate 568 part 1, and checkmate 012 (arms p and q).
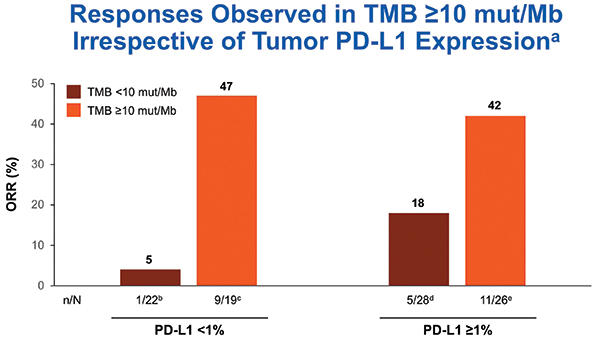
Checkmate 568 Efficacy And Biomarker Analysis For Nivolumab And Here, we report the efficacy of first line nivolumab plus ipilimumab treatment in a large, pooled population of patients with advanced nsclc from checkmate 227 part 1, checkmate 817 (cohort a), checkmate 568 part 1, and checkmate 012 (arms p and q).

Pdf Biomarker Analysis From Checkmate 214 Nivolumab Plus Ipilimumab
Comments are closed.