Chapter Bonding Pdf

Chapter 13 Bonding Pdf Understand, in simple terms, the concept of electronegativity and apply it to explain the properties of molecules such as bond polarity and the dipole moments of molecules. Find the sum of valence electrons of all atoms in the polyatomic ion or molecule. if it is an anion, add one electron for each negative charge.

Bonding Pdf The central importance of chemical bonding to the subject is well recognised…, and is such that it is considered a core topic in many chemistry curricula at school, college and university level. Bonding energies and forces forces can be calculated from the potential energy of inter atomic interaction. Bonds, called covalent bonds (latin co valere for joint strength), are not limited to homonuclear molecules but also occur between atoms of different element. A covalent bond is a chemical bond where electron pairs are shared between atoms. a covalent bond is the strong electrostatic attraction between a shared pair of electrons and the nuclei of the bonding atoms.

Chapter 8 Covalent Bonding Study Guide Mcgraw Hill Doc Template Bonds, called covalent bonds (latin co valere for joint strength), are not limited to homonuclear molecules but also occur between atoms of different element. A covalent bond is a chemical bond where electron pairs are shared between atoms. a covalent bond is the strong electrostatic attraction between a shared pair of electrons and the nuclei of the bonding atoms. Chemical bonding full notes pdf free download as pdf file (.pdf), text file (.txt) or read online for free. the document discusses chemical bonding and lewis structures. Mastery of the following concepts discussed in chapter 1 is essential for all aspects of understanding organic chemistry: (1)covalent bonds and lewis structures. lewis structures are the "words and grammar" of the language of organic chemistry. Each bond classification is discussed in detail in subsequent sections of the chapter. let's look at the preferred arrangements of electrons in atoms when they form chemical compounds. Write lewis symbols for atoms and ions. define lattice energy and be able to arrange compounds in order of increasing lattice energy based on the charges and sizes of the ions involved. use atomic electron configurations and the octet rule to draw lewis structures for molecules.
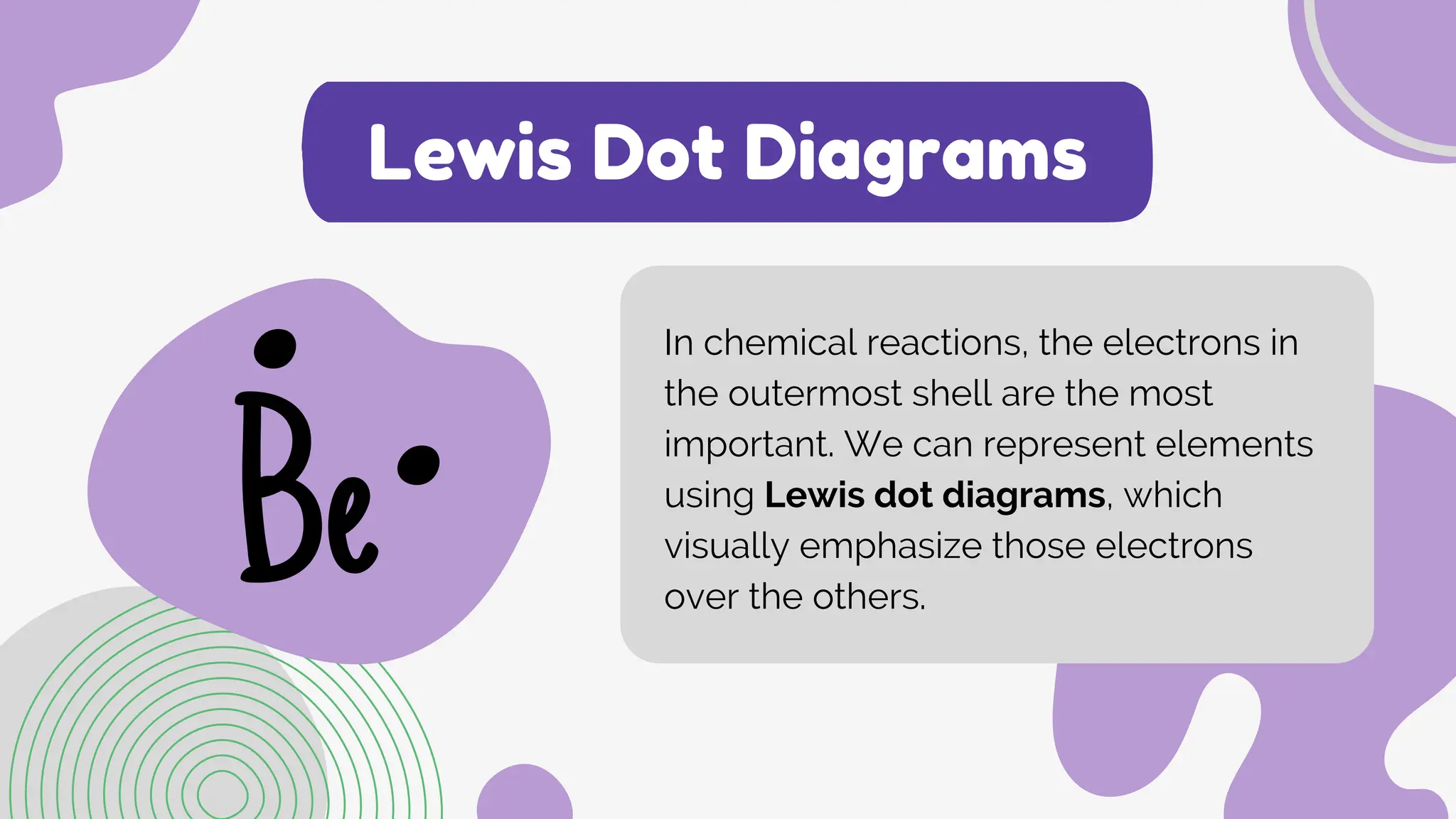
Introduction Of Chemical Bonding Pdf Chemical bonding full notes pdf free download as pdf file (.pdf), text file (.txt) or read online for free. the document discusses chemical bonding and lewis structures. Mastery of the following concepts discussed in chapter 1 is essential for all aspects of understanding organic chemistry: (1)covalent bonds and lewis structures. lewis structures are the "words and grammar" of the language of organic chemistry. Each bond classification is discussed in detail in subsequent sections of the chapter. let's look at the preferred arrangements of electrons in atoms when they form chemical compounds. Write lewis symbols for atoms and ions. define lattice energy and be able to arrange compounds in order of increasing lattice energy based on the charges and sizes of the ions involved. use atomic electron configurations and the octet rule to draw lewis structures for molecules.

Bonding Pdf Each bond classification is discussed in detail in subsequent sections of the chapter. let's look at the preferred arrangements of electrons in atoms when they form chemical compounds. Write lewis symbols for atoms and ions. define lattice energy and be able to arrange compounds in order of increasing lattice energy based on the charges and sizes of the ions involved. use atomic electron configurations and the octet rule to draw lewis structures for molecules.

Chapter A1 Bonding Pdf
Comments are closed.