Chapter 6 Functional Groups Pdf Alkane Methyl Group

Alkane Group Pdf Alkane Propane 1) functional groups are specific groupings of atoms that have characteristic properties regardless of other atoms in a molecule. common examples include alcohols, alkenes, alkynes, amines, carboxylic acids, aldehydes, and ketones. The simplest functional group in organic chemistry (which is often ignored when listing functional groups) is called an alkane, characterized by single bonds between two carbons and between carbon and hydrogen.

Chapter 6 Functional Groups Pdf Alkane Methyl Group The iso structural unit consists of two methyl groups attached to a common carbon. when this unit is present in an alkane or alky group, the common name starts with the prefix iso. Introduction: alkanes are organic compounds that consist entirely of single bonded carbon and hydrogen atoms and lack any other functional groups. alkanes have the general formula cnh2n 2, alkanes, also called paraffin’s. • organic molecules have a skeleton of carbon atoms, often with functional groups attached. • hydrocarbons (carbons & hydrogens only) have 2 classes: saturated alkanes & unsaturated alkenes, alkynes & arenes. In a later portion of this chapter, you will learn how to systematically name compounds by counting the number of carbons in the longest continuous chain and identifying any functional groups present.
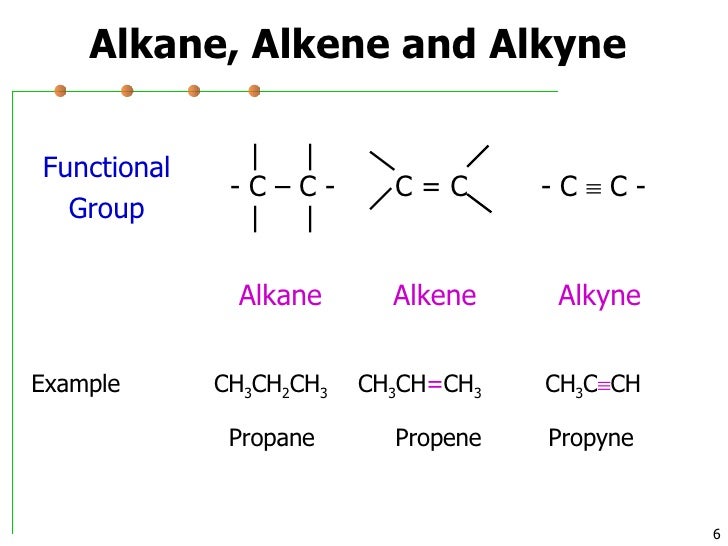
Alkane Functional Group • organic molecules have a skeleton of carbon atoms, often with functional groups attached. • hydrocarbons (carbons & hydrogens only) have 2 classes: saturated alkanes & unsaturated alkenes, alkynes & arenes. In a later portion of this chapter, you will learn how to systematically name compounds by counting the number of carbons in the longest continuous chain and identifying any functional groups present. Alkane substituents are called alkyl groups, which refers to alkane groups lacking a c h bond such as methyl, ethyl, or propyl. common examples of alkanes are methane, ethane, propane, butane, and octane. Alkene (c=c) nomenclature drop the “ ane” suffix of alkane and add ene (ending) eg. propane > propene. the longest chain chosen for the root name must include both carbon atoms of the double bond. the root chain must be numbered from the end nearest a double bond carbon atom. The chemistry of the organic molecule is defined by the function groups it contains. Since the ability to recognize these functional groups, particularly when they are incorporated into a larger, more complicated molecule, is so important, you should learn the names and general structures of all of the functional groups on this handout as soon as possible.

Free Functional Group Alkane Icons Symbols Images Biorender Alkane substituents are called alkyl groups, which refers to alkane groups lacking a c h bond such as methyl, ethyl, or propyl. common examples of alkanes are methane, ethane, propane, butane, and octane. Alkene (c=c) nomenclature drop the “ ane” suffix of alkane and add ene (ending) eg. propane > propene. the longest chain chosen for the root name must include both carbon atoms of the double bond. the root chain must be numbered from the end nearest a double bond carbon atom. The chemistry of the organic molecule is defined by the function groups it contains. Since the ability to recognize these functional groups, particularly when they are incorporated into a larger, more complicated molecule, is so important, you should learn the names and general structures of all of the functional groups on this handout as soon as possible.

Functional Groups Pdf Functional Group Alkane The chemistry of the organic molecule is defined by the function groups it contains. Since the ability to recognize these functional groups, particularly when they are incorporated into a larger, more complicated molecule, is so important, you should learn the names and general structures of all of the functional groups on this handout as soon as possible.
Comments are closed.