Chapter 5 Diffusion Part 1

Chapter 2 Molecular Diffusion Part 1 Pdf Diffusion Heat Issues to address • how does diffusion occur? • why is it an important part of processing? • how can the rate of diffusion be predicted for some simple cases? • how does diffusion depend on structure and temperature?. One practical example of steady state diffusion is found in the purification of hydrogen gas. one side of a thin sheet of palladium metal is exposed to the impure gas composed of hydrogen and other gaseous species such as nitrogen, oxygen, and water vapor.

Chapter 5 Diffusion Flashcards Quizlet Chapter 5 discusses the phenomenon of diffusion in solids, highlighting its significance in material processing, particularly during heat treatment. it covers various diffusion mechanisms, including interdiffusion and self diffusion, and explains how diffusion rates can be predicted using fick's laws. Chapter 5: diffusion (1) diffusion mass transport by random, atomic (or molecular scale) motion. View lecture slides diffusion.pdf from mechanical 256 at Özyeğin university. chapter 5: diffusion issues to address. • how does diffusion occur? • why is it an important part of processing? • how. Question: consider the self diffusion of two hypothetical metals a and b. on a schematic graph of ln d versus 1 t, plot (and label) lines for both metals given that d0(a) > d0(b) and also that qd(a) > qd(b).

Ppt Chapter 5 Diffusion In Solids Powerpoint Presentation Free View lecture slides diffusion.pdf from mechanical 256 at Özyeğin university. chapter 5: diffusion issues to address. • how does diffusion occur? • why is it an important part of processing? • how. Question: consider the self diffusion of two hypothetical metals a and b. on a schematic graph of ln d versus 1 t, plot (and label) lines for both metals given that d0(a) > d0(b) and also that qd(a) > qd(b). Diffusion – how do atoms move through solids? diffusion is material transport by atomic motion. inhomogeneous materials can become homogeneous by diffusion. for an active diffusion to occur, the temperature should be high enough to overcome energy barriers to atomic motion. Interstitial diffusion is generally faster than vacancy diffusion because bonding of interstitials to the surrounding atoms is normally weaker and there are many more interstitial sites than vacancy sites to jump to. requires small impurity atoms (e.g. c, h, o) to fit into interstices in host. Explore the principles of diffusion in materials, including mechanisms, mathematical modeling, and applications in heat treatment processes. Diffusion demo glass tube filled with water. at time t = 0, add some drops of ink to one end of the tube. measure the diffusion distance, x, over some time. compare the results with theory.

Ppt Chapter 5 Diffusion Powerpoint Presentation Free Download Id Diffusion – how do atoms move through solids? diffusion is material transport by atomic motion. inhomogeneous materials can become homogeneous by diffusion. for an active diffusion to occur, the temperature should be high enough to overcome energy barriers to atomic motion. Interstitial diffusion is generally faster than vacancy diffusion because bonding of interstitials to the surrounding atoms is normally weaker and there are many more interstitial sites than vacancy sites to jump to. requires small impurity atoms (e.g. c, h, o) to fit into interstices in host. Explore the principles of diffusion in materials, including mechanisms, mathematical modeling, and applications in heat treatment processes. Diffusion demo glass tube filled with water. at time t = 0, add some drops of ink to one end of the tube. measure the diffusion distance, x, over some time. compare the results with theory.
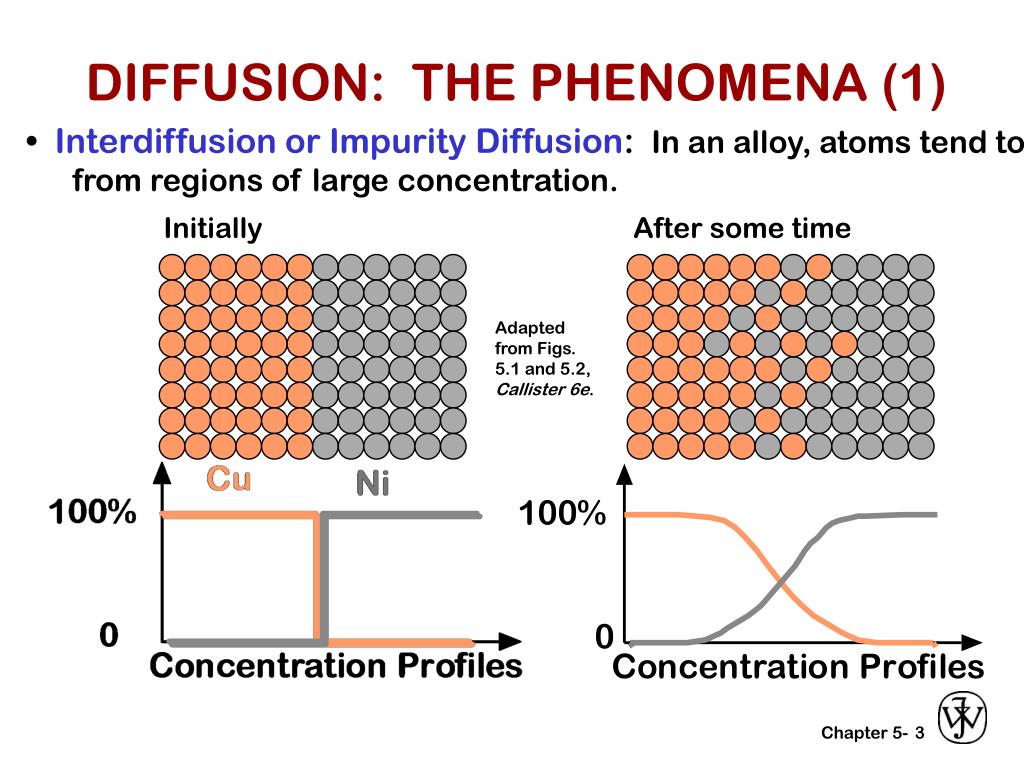
Ppt Chapter 5 Diffusion In Solids Powerpoint Presentation Free Explore the principles of diffusion in materials, including mechanisms, mathematical modeling, and applications in heat treatment processes. Diffusion demo glass tube filled with water. at time t = 0, add some drops of ink to one end of the tube. measure the diffusion distance, x, over some time. compare the results with theory.

Solution Chapter 5 Module 1 The Diffusion Process 1 Studypool
Comments are closed.