Cell Notation Practice Problems Voltaic Cells Electrochemistry

Cell Notation Practice Problems Voltaic Cells Electrochemistry In one episode of the tv sitcom, gilligan’s island, the “professor” constructed voltaic cells to use as substitutes for their radio’s dead batteries. which scraps of metal from their damaged boat, the minnow, could best be used to create a 1.5 v voltaic cell?. Electrochemistry practice problems covering voltaic cells, cell potentials, redox reactions, and electrolysis. ideal for chemistry students.

Voltaic Cell Worksheet Pdf Electrode Electrochemistry If you missed any regents practice questions you should see me for extra help and or re watch the lesson video assignment 17. Electrochemistry practice problems include questions on calculating eo and e, Δg based on the cell potential, nernst equation, and more. 1. writing cell notation cells are formed from the following half cells. write the notation for the cells. Explore cell notation with interactive practice questions. get instant answer verification, watch video solutions, and gain a deeper understanding of this essential general chemistry topic.

Practice Problems Electrochemical Cell Pdf Electrochemistry Anode 1. writing cell notation cells are formed from the following half cells. write the notation for the cells. Explore cell notation with interactive practice questions. get instant answer verification, watch video solutions, and gain a deeper understanding of this essential general chemistry topic. Write cell reactions for the electrochemical cells diagrammed, and use data from the table of standard electrode potentials to calculate \ (\mathrm {e^\circ {cell}}\) for each reaction. Calculate the standard cell potential produced by a voltaic cell consisting of a gold electrode in contact with a solution of au3 ions and a silver electrode in contact with a solution of ag ions. Students can use this free online quiz to practice electrochemistry calculations for a variety of cell potentials and conditions. Given the following half reactions and half cell potentials, write the balanced overall electrochemical reaction that would occur and calculate the cell potential of a voltaic cell incorporating these two half reactions.

Electrochemistry Voltaic Cell Practice Worksheet By The Scientific Write cell reactions for the electrochemical cells diagrammed, and use data from the table of standard electrode potentials to calculate \ (\mathrm {e^\circ {cell}}\) for each reaction. Calculate the standard cell potential produced by a voltaic cell consisting of a gold electrode in contact with a solution of au3 ions and a silver electrode in contact with a solution of ag ions. Students can use this free online quiz to practice electrochemistry calculations for a variety of cell potentials and conditions. Given the following half reactions and half cell potentials, write the balanced overall electrochemical reaction that would occur and calculate the cell potential of a voltaic cell incorporating these two half reactions.
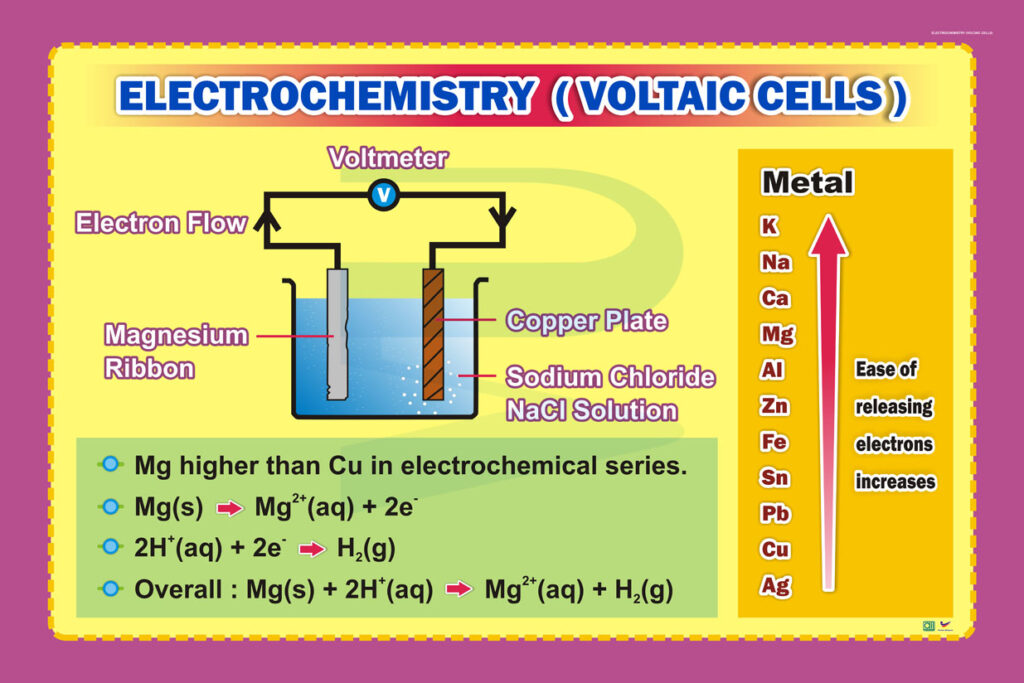
Electrochemistry Voltaic Cells Progressive Scientific Sdn Bhd Students can use this free online quiz to practice electrochemistry calculations for a variety of cell potentials and conditions. Given the following half reactions and half cell potentials, write the balanced overall electrochemical reaction that would occur and calculate the cell potential of a voltaic cell incorporating these two half reactions.
Comments are closed.