Catalyst And Its Types Pdf Notes

Catalyst Pdf Catalysis Chemical Process Engineering There are two main types of catalysis: homogeneous catalysis where reactants and catalysts are in the same phase, and heterogeneous catalysis where they are in different phases. A catalyst is chosen that releases the products formed as readily as possible; otherwise the products remain on the catalyst surface and act as poisons to the process.

Catalyst Pdf Catalysis Nanoparticle Energy. in autocatalysis a reaction product is itself a catalyst for that reaction leading to positive feedback. proteins that act as catalysts in biochemical reactions are called enzymes. a catalyst does not affect the position of the equilibrium, as the catalyst speeds up the backward and forward reactions equally. Catalysis: (a) types of catalysis, heterogeneous and homogenous catalysis, advantages and disadvantages 41 (b) heterogeneous catalysis – preparation, characterization, kinetics, supported. There are many different types of catalysts. they range from the proton, h , through lewis acids, organometallic complexes, organic and inorganic polymers, all the way to enzymes. Reaction rate is called a catalyst. catalysts work by providing alternative mechanism involving a differe t transition state of lower energy. thereby, the activation energy of the catalytic reaction is lowered compared to the unc.

Catalysis And Types Of Catalysts Pdf Catalysis Heterogeneous There are many different types of catalysts. they range from the proton, h , through lewis acids, organometallic complexes, organic and inorganic polymers, all the way to enzymes. Reaction rate is called a catalyst. catalysts work by providing alternative mechanism involving a differe t transition state of lower energy. thereby, the activation energy of the catalytic reaction is lowered compared to the unc. What is catalysis? a catalyst accelerates a chemical reaction. it does so by forming bonds with the reacting molecules, and by allowing these to react to a product, which detaches from the catalyst, and leaves it unaltered such that it is available for the next reaction. Lecture notes about catalysis. inorganic and enzyme catalysts and their properties; kinetics of heterogeneous catalytic reactions; adsorption isotherms, derivation of rate laws; langmuir hinshelwood kinetics. Homogeneous catalysis: when the reactants and the catalyst are in the same phase (i.e. solid, liquid or gas). the catalysis is said to be homogeneous. the following are some of the examples of homogeneous catalysis. Questions for self control define catalysis and explain how catalysts increase reaction rates. what are the main differences between homogeneous and heterogeneous catalysis? describe the steps involved in heterogeneous catalytic reactions. write examples of industrial processes using solid catalysts.

Catalyst Catalysis Types Of Catalyst And Catalysis Pptx Chemistry What is catalysis? a catalyst accelerates a chemical reaction. it does so by forming bonds with the reacting molecules, and by allowing these to react to a product, which detaches from the catalyst, and leaves it unaltered such that it is available for the next reaction. Lecture notes about catalysis. inorganic and enzyme catalysts and their properties; kinetics of heterogeneous catalytic reactions; adsorption isotherms, derivation of rate laws; langmuir hinshelwood kinetics. Homogeneous catalysis: when the reactants and the catalyst are in the same phase (i.e. solid, liquid or gas). the catalysis is said to be homogeneous. the following are some of the examples of homogeneous catalysis. Questions for self control define catalysis and explain how catalysts increase reaction rates. what are the main differences between homogeneous and heterogeneous catalysis? describe the steps involved in heterogeneous catalytic reactions. write examples of industrial processes using solid catalysts.
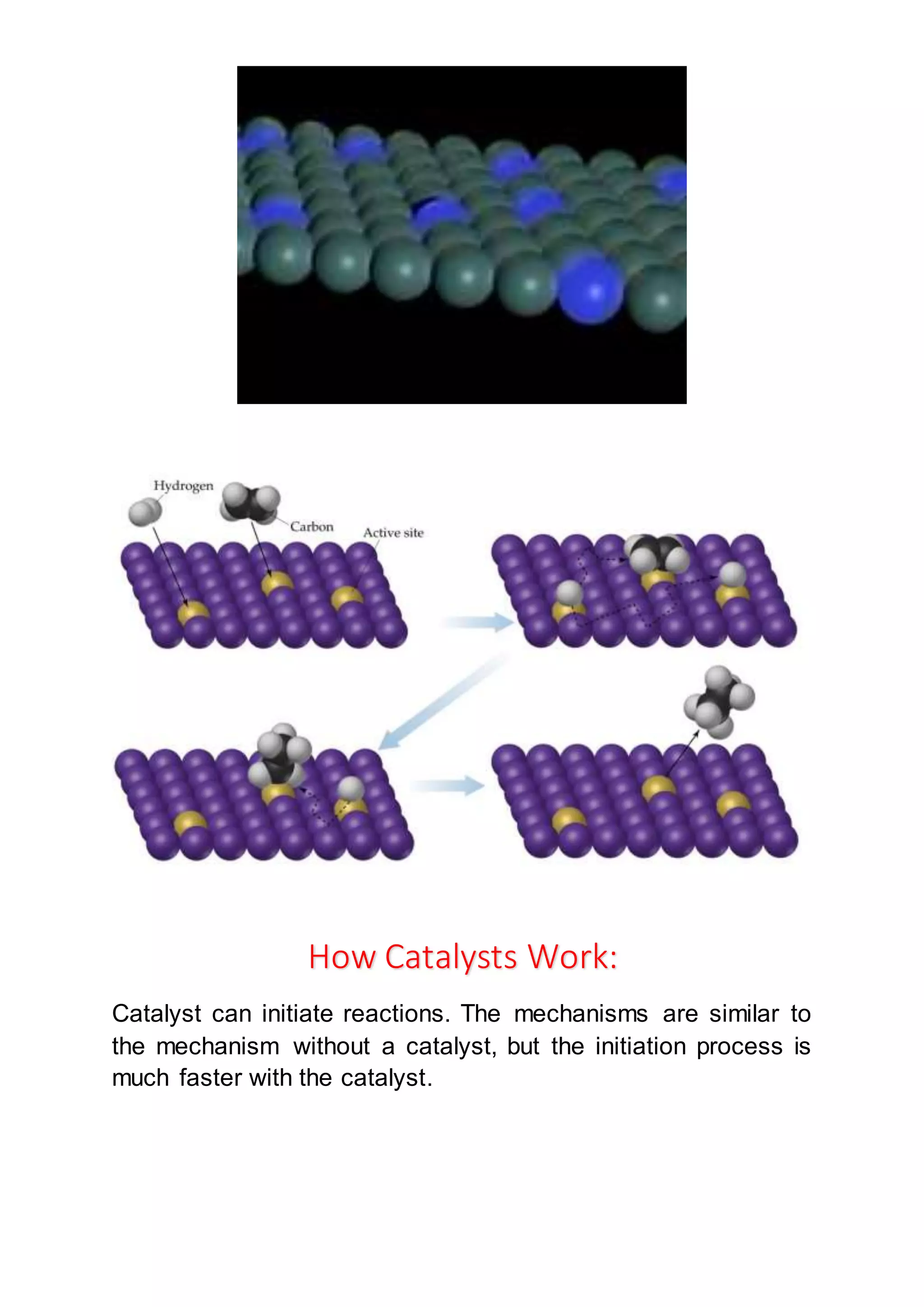
Catalyst And Their Types Docx Homogeneous catalysis: when the reactants and the catalyst are in the same phase (i.e. solid, liquid or gas). the catalysis is said to be homogeneous. the following are some of the examples of homogeneous catalysis. Questions for self control define catalysis and explain how catalysts increase reaction rates. what are the main differences between homogeneous and heterogeneous catalysis? describe the steps involved in heterogeneous catalytic reactions. write examples of industrial processes using solid catalysts.

02b Principles Of Catalysis Annotated Notes 2313spr2018 Pdf
Comments are closed.