Catalysis Types And Characteristics Pdf

3 Catalysis And Types Pdf Catalysis: (a) types of catalysis, heterogeneous and homogenous catalysis, advantages and disadvantages 41 (b) heterogeneous catalysis – preparation, characterization, kinetics,. A catalyst is chosen that releases the products formed as readily as possible; otherwise the products remain on the catalyst surface and act as poisons to the process.

Catalysis And Types Of Catalysts Pdf Catalysis Heterogeneous What is catalysis? a catalyst accelerates a chemical reaction. it does so by forming bonds with the reacting molecules, and by allowing these to react to a product, which detaches from the catalyst, and leaves it unaltered such that it is available for the next reaction. These catalysts were originated in the 1950s by the german chemist karl ziegler for the polymerization of ethylene at atmospheric pressure. ziegler employed a catalyst consisting of a mixture of titanium tetrachloride and an alkyl derivative of aluminum. Understanding catalysis: types & characteristics this document discusses different types of catalysis: [1] homogeneous catalysis involves reactants and catalysts in the same physical state. We will start the unit by defining catalysis and the terminology used in the context of catalysis. we would then classify catalysis and outline its characteristics. this will be followed by a discussion on homogeneous catalysis.

Catalysis 3 Pdf Understanding catalysis: types & characteristics this document discusses different types of catalysis: [1] homogeneous catalysis involves reactants and catalysts in the same physical state. We will start the unit by defining catalysis and the terminology used in the context of catalysis. we would then classify catalysis and outline its characteristics. this will be followed by a discussion on homogeneous catalysis. Velopment in the chemical industry. catalysis involves understanding of the thermodynamics, kinetics, electronic interaction, crystal structure, reactor design and process . 1.1.2 heterogeneous catalysis: the relationship between a catalyst's performance and its composition and structure 4. Although the catalysts and the process conditions in each category can be very different, the principles of catalysis are the same. once you understand these principles, you can apply them in all three categories. Homogeneous catalysis: when the reactants and the catalyst are in the same phase (i.e. solid, liquid or gas). the catalysis is said to be homogeneous. the following are some of the examples of homogeneous catalysis.

An Introduction To Types Of Catalysis Download Free Pdf Catalysis Velopment in the chemical industry. catalysis involves understanding of the thermodynamics, kinetics, electronic interaction, crystal structure, reactor design and process . 1.1.2 heterogeneous catalysis: the relationship between a catalyst's performance and its composition and structure 4. Although the catalysts and the process conditions in each category can be very different, the principles of catalysis are the same. once you understand these principles, you can apply them in all three categories. Homogeneous catalysis: when the reactants and the catalyst are in the same phase (i.e. solid, liquid or gas). the catalysis is said to be homogeneous. the following are some of the examples of homogeneous catalysis.
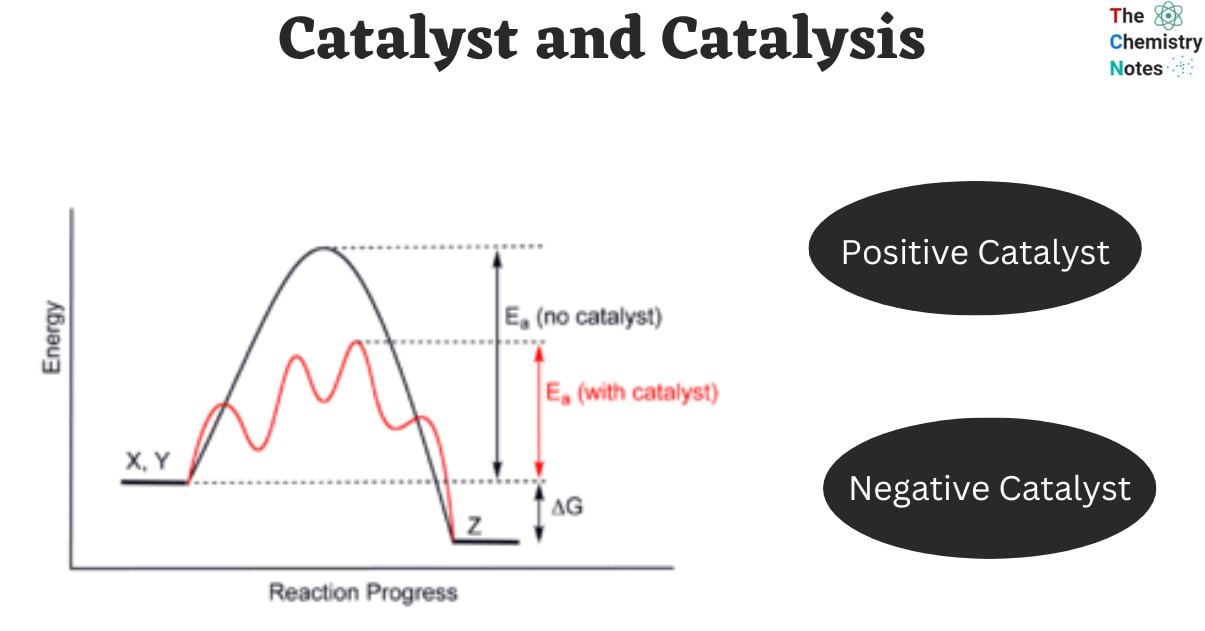
Catalyst And Catalysis Types Examples Differences Although the catalysts and the process conditions in each category can be very different, the principles of catalysis are the same. once you understand these principles, you can apply them in all three categories. Homogeneous catalysis: when the reactants and the catalyst are in the same phase (i.e. solid, liquid or gas). the catalysis is said to be homogeneous. the following are some of the examples of homogeneous catalysis.

Catalysis Types Of Catalysis For Chemistry Students Ppt
Comments are closed.