Carbenes Carbene Stabilisation

Carbene Introduction Singlet And Triplet Carbenes Preparation Of To facilitate the design of broader classes of enzymes that can take advantage of the rich carbene chemistry, it is thus important to better understand how to stabilize carbene species in enzyme active sites without metal ions. Nowadays, aside from the carbenes mentioned above, a myriad of stable carbenes with distinct steric and electronic properties are available.
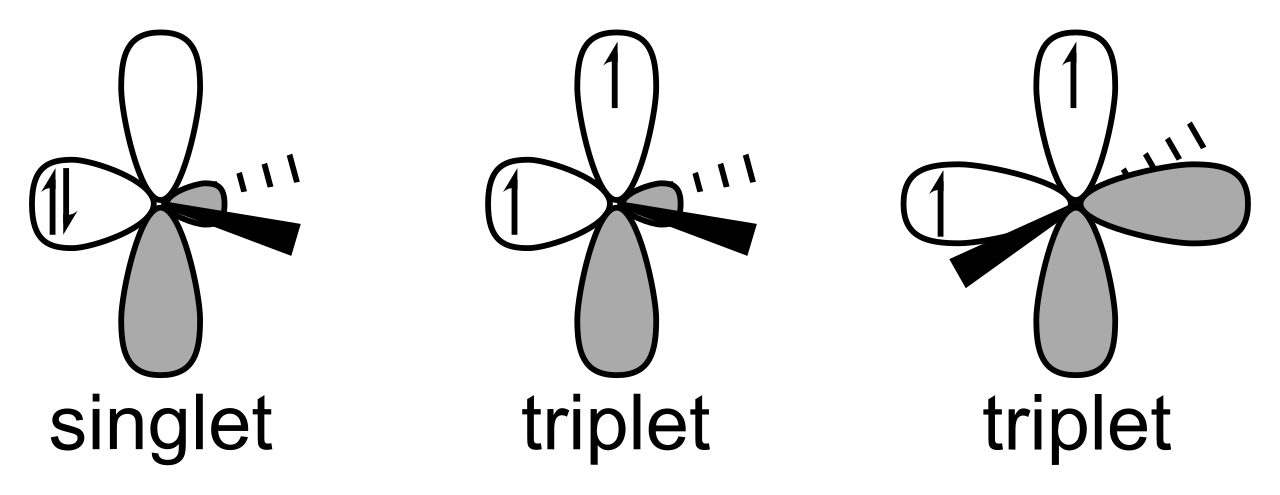
Carbene Chemtalk If there are more than one donor atoms attached to the carbene that can become appropriately aligned, further stability is possible. the first stable carbene isolated was a n heterocyclic carbene. This chapter begins with a brief discussion of strategies for assessing carbene stability and the experimental and computational methods that have been used. the chapter then moves directly to the stabilities of various classes of carbenes. Carbenes are divalent carbon intermediates containing a carbon atom with only six valence electrons and two unshared electrons. they are highly reactive and play an important role in many organic reactions, especially in cyclopropanation, rearrangements, and insertions. The reactivity of carbenes is somewhat reduced when stabilization results from the phenomenon known as resonance, which is said to occur when alternate electronic structures (called resonance forms) can be written for a single molecular type.

Carbene Chemtalk Carbenes are divalent carbon intermediates containing a carbon atom with only six valence electrons and two unshared electrons. they are highly reactive and play an important role in many organic reactions, especially in cyclopropanation, rearrangements, and insertions. The reactivity of carbenes is somewhat reduced when stabilization results from the phenomenon known as resonance, which is said to occur when alternate electronic structures (called resonance forms) can be written for a single molecular type. The ways of solving the problem of stability of carbenes by the methods of quantum chemistry are discussed, in particular the application of new criteria for the stabilization of cyclic. We have summarized the arylation, vinylation, and alkylation of carbenes utilizing various transition metals, viz. palladium, rhodium, copper, and platinum, for the construction of carbon–carbon bonds, carbon–boron bonds, and beyond through the cross coupling strategy. Catalytic c h activation: carbenes can serve as catalysts for selective c h bond activation, enabling functionalization of inert c h bonds in complex molecules. Carbenes, including n ‐heterocyclic carbene (nhc) ligands, are used extensively to stabilize open‐shell transition metal complexes and organic radicals. yet, it remains unknown, which carbene stabilizes a radical well and, thus, how to design radical‐stabilizing c‐donor ligands.

Carbene And Nitrene Cyclization Carbenes Are Uncharged Electron The ways of solving the problem of stability of carbenes by the methods of quantum chemistry are discussed, in particular the application of new criteria for the stabilization of cyclic. We have summarized the arylation, vinylation, and alkylation of carbenes utilizing various transition metals, viz. palladium, rhodium, copper, and platinum, for the construction of carbon–carbon bonds, carbon–boron bonds, and beyond through the cross coupling strategy. Catalytic c h activation: carbenes can serve as catalysts for selective c h bond activation, enabling functionalization of inert c h bonds in complex molecules. Carbenes, including n ‐heterocyclic carbene (nhc) ligands, are used extensively to stabilize open‐shell transition metal complexes and organic radicals. yet, it remains unknown, which carbene stabilizes a radical well and, thus, how to design radical‐stabilizing c‐donor ligands.

Carbene Footprinting Photene Catalytic c h activation: carbenes can serve as catalysts for selective c h bond activation, enabling functionalization of inert c h bonds in complex molecules. Carbenes, including n ‐heterocyclic carbene (nhc) ligands, are used extensively to stabilize open‐shell transition metal complexes and organic radicals. yet, it remains unknown, which carbene stabilizes a radical well and, thus, how to design radical‐stabilizing c‐donor ligands.
Comments are closed.