Cancer Treatment Inflames Cold Tumors For Heat Seeking Immune Cells

Cancer Treatment Inflames Cold Tumors For Heat Seeking Immune Cells In patients with solid tumors, those who respond well to icb treatment typically demonstrate an active immune profile referred to as the “hot” (immune inflamed) phenotype. This facilitates robust immune cell infiltration and effector function, transforming immune excluded tumors into t cell inflamed microenvironments to potentiate immunotherapy responses.

Making Immune Cold Tumors Hot Bioserendipity The revolution in cancer immunotherapy, particularly through immune checkpoint inhibitors (icis), underscores the significant role of the tumor microenvironment (tme) in determining therapeutic outcomes. Combining immunotherapy with a range of treatment modalities allow acting on different immune cells and improve immune response in the clinical setting. we review the current strategies to inflame cold tumors that are currently being tested in clinical trials for multiple indications. In this review, we summarize the intrinsic, extrinsic, and systemic factors contributing to the formation of ‘cold’ tumors within the framework of the cancer immunity cycle. correspondingly, we discuss potential strategies for converting ‘cold’ tumors into ‘hot’ ones to enhance therapeutic efficacy. In ‘cold’ tumors, the absence of t cell infiltration leads to the ineffectiveness of ici therapy. addressing these challenges, especially the impairment in t cell activation and homing, is crucial to enhance ici therapy’s efficacy.
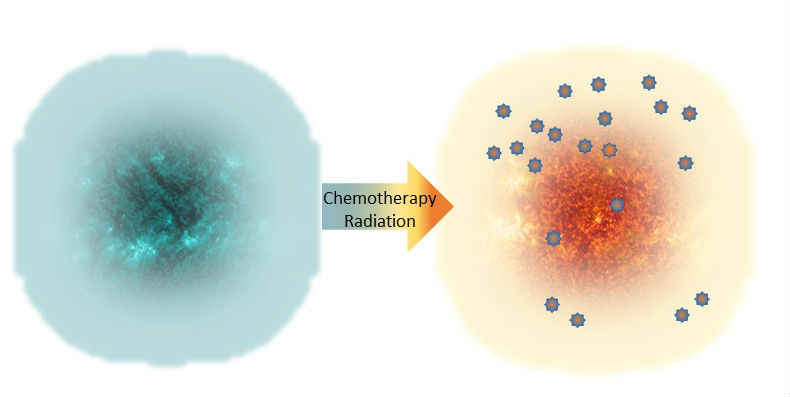
Making Immune Cold Tumors Hot Bioserendipity In this review, we summarize the intrinsic, extrinsic, and systemic factors contributing to the formation of ‘cold’ tumors within the framework of the cancer immunity cycle. correspondingly, we discuss potential strategies for converting ‘cold’ tumors into ‘hot’ ones to enhance therapeutic efficacy. In ‘cold’ tumors, the absence of t cell infiltration leads to the ineffectiveness of ici therapy. addressing these challenges, especially the impairment in t cell activation and homing, is crucial to enhance ici therapy’s efficacy. This review sheds light on current advancements and future directions in the quest for more effective and safer cancer treatment strategies, offering hope for patients with immune resistant tumors. This study explores how to convert immune cold tumors into immune responsive or “immune hot” tumors, enabling immune cells such as b cells and t cells to attack cancer cells more effectively and enhance the success of chemotherapy and immunotherapy. This review will provide a brief summary of different tumor properties contributing to the establishment of cold tumors and describe major strategies that could reprogram non inflamed cold tumors into inflamed hot tumors. Herein, we systematically outlined the mechanisms underlying the formation of the immunosuppressive tme in cold tumors, including the role of immunosuppressive cells, impaired antigen presentation, transforming growth factor β, stat3 signaling, adenosine, and interferon γ signaling.
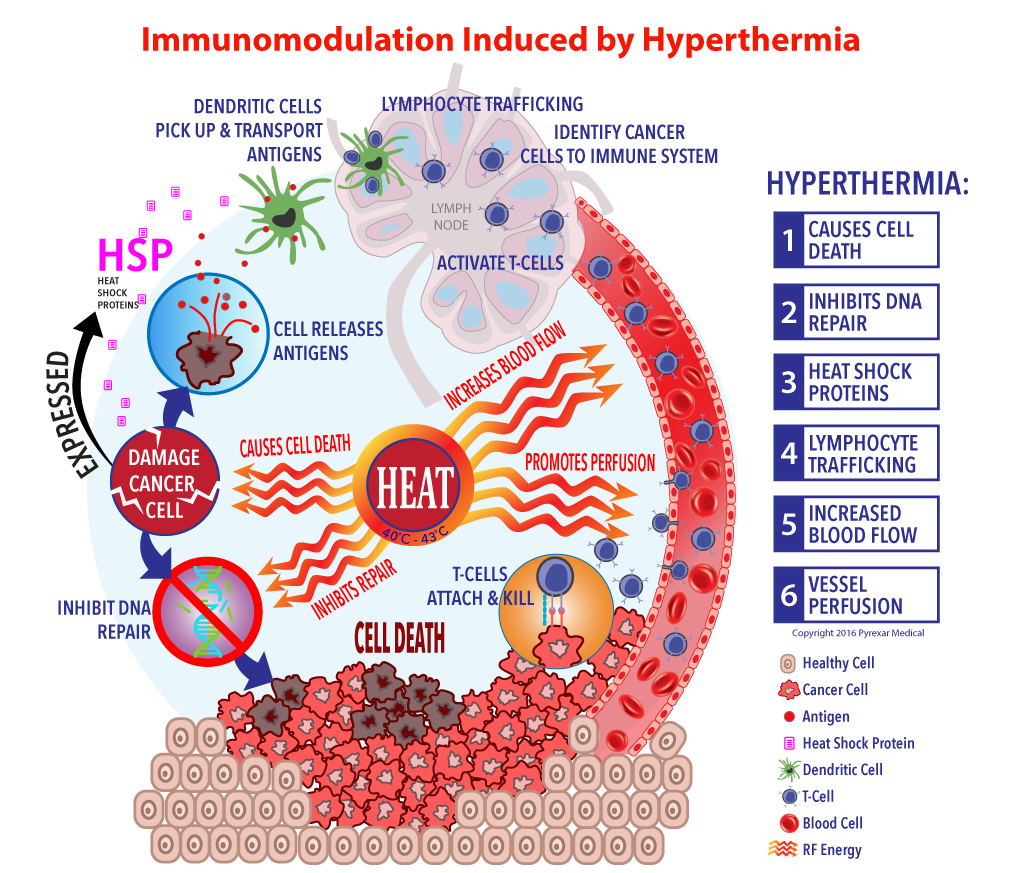
Heat Kills Cancer Tumors At Daniel Shears Blog This review sheds light on current advancements and future directions in the quest for more effective and safer cancer treatment strategies, offering hope for patients with immune resistant tumors. This study explores how to convert immune cold tumors into immune responsive or “immune hot” tumors, enabling immune cells such as b cells and t cells to attack cancer cells more effectively and enhance the success of chemotherapy and immunotherapy. This review will provide a brief summary of different tumor properties contributing to the establishment of cold tumors and describe major strategies that could reprogram non inflamed cold tumors into inflamed hot tumors. Herein, we systematically outlined the mechanisms underlying the formation of the immunosuppressive tme in cold tumors, including the role of immunosuppressive cells, impaired antigen presentation, transforming growth factor β, stat3 signaling, adenosine, and interferon γ signaling.

Cancer Immunotherapy Could Turn Up The Heat On Cold Tumors By This review will provide a brief summary of different tumor properties contributing to the establishment of cold tumors and describe major strategies that could reprogram non inflamed cold tumors into inflamed hot tumors. Herein, we systematically outlined the mechanisms underlying the formation of the immunosuppressive tme in cold tumors, including the role of immunosuppressive cells, impaired antigen presentation, transforming growth factor β, stat3 signaling, adenosine, and interferon γ signaling.
Comments are closed.