Calorimetry Lab 3 0 Overview

Calorimetry Theory Module 3 Pdf Carter shows us how to use the new layout and features in the new web based calorimetry lab. Calorimetry is based on the first law of thermodynamics that states that energy cannot be created nor destroyed. the heat of neutralization that is lost in the chemical reaction (the system) is gained by the calorimeter and its contents (the surroundings).
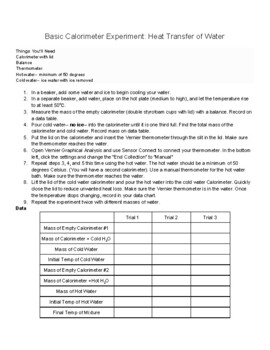
Calorimetry Lab Basic By The Chem And Physics Lady Tpt Calorimetry is a collection of experimental techniques used to measure energy changes and heat flows associated with chemical reactions and physical changes. the term "calorimetry" comes from the greek words for heat ("calor") and measure ("meter"). Embark on a fascinating journey into the world of thermal science with our student exploration calorimetry lab guide. this experiment allows you to determine the heat transferred during physical and chemical changes. we will use the principles of thermodynamics to understand energy flow. What is calorimetry? calorimetry is a branch of science concerned with measuring a body’s state in terms of thermal features to investigate its physical and chemical changes. to determine the enthalpy, stability, heat capacity, and other thermochemical quantities, calorimetry is widely used. Introduction in this lab, you will use knowledge regarding thermochemistry and calorimetry. calorimetry is a technique where the amount of heat involved in a chemical or physical process is measured in a closed environment.
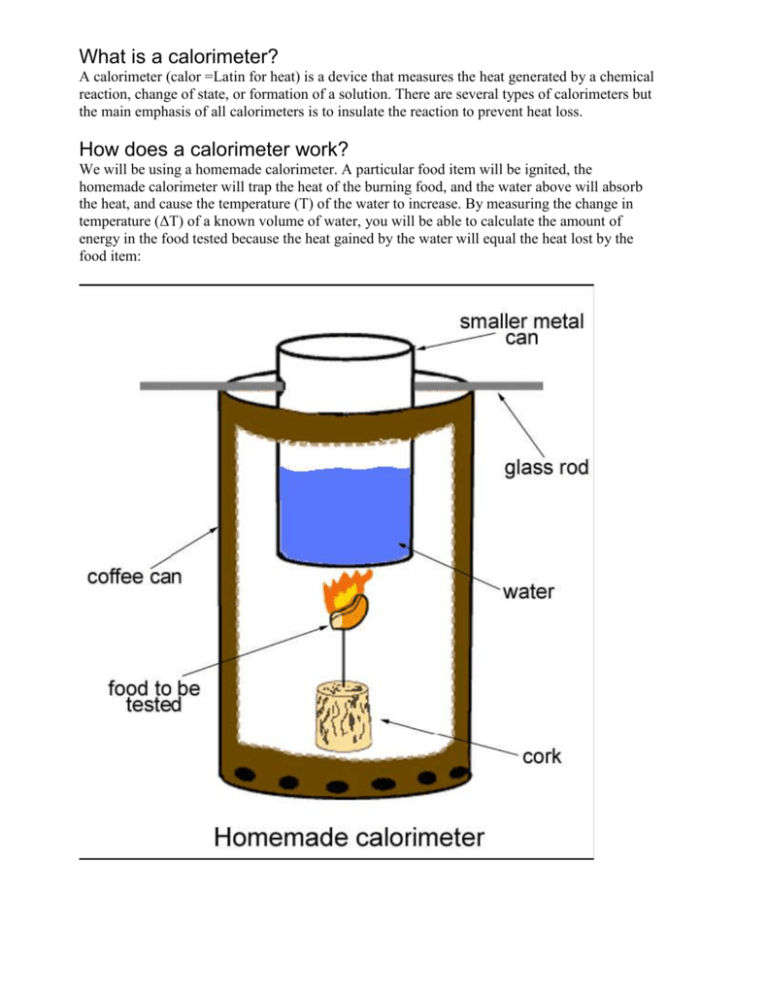
Introduction To Calorimetry Lab Report At Bob Wright Blog What is calorimetry? calorimetry is a branch of science concerned with measuring a body’s state in terms of thermal features to investigate its physical and chemical changes. to determine the enthalpy, stability, heat capacity, and other thermochemical quantities, calorimetry is widely used. Introduction in this lab, you will use knowledge regarding thermochemistry and calorimetry. calorimetry is a technique where the amount of heat involved in a chemical or physical process is measured in a closed environment. Learn how calorimetry is used in thermochemistry to measure heat transferred during chemical reactions, phase changes, and specific heat experiments. Calorimetry is the process of measuring heat changes (released or absorbed) during a chemical reaction or a physical phenomenon. those changes are measured by a calorimeter that consists of a container with walls kept at constant temperature and in which the thermal phenomenon is investigated. Explore calorimetry experiments in ib chemistry. learn how to calculate energy changes from temperature, mass, and specific heat capacity. In this experiment you will become familiar with the basic principles of calorimetry. you will use a bomb calorimeter, in which chemical reactions take place without change of volume (isochoric reaction path). the tem perature change in a surrounding bath is monitored.

Calorimetry Lab Report Phys1006 Foundations Of Physics Thinkswap Learn how calorimetry is used in thermochemistry to measure heat transferred during chemical reactions, phase changes, and specific heat experiments. Calorimetry is the process of measuring heat changes (released or absorbed) during a chemical reaction or a physical phenomenon. those changes are measured by a calorimeter that consists of a container with walls kept at constant temperature and in which the thermal phenomenon is investigated. Explore calorimetry experiments in ib chemistry. learn how to calculate energy changes from temperature, mass, and specific heat capacity. In this experiment you will become familiar with the basic principles of calorimetry. you will use a bomb calorimeter, in which chemical reactions take place without change of volume (isochoric reaction path). the tem perature change in a surrounding bath is monitored.
Comments are closed.