Calorimetry Grade12uchemistry
Calorimetry Chemtalk The presentation models the solving of calorimetry calculations. explain the use of a calorimeter and have students work on an inquiry activity using a coffee cup calorimeter. Grade 12 calorimetry worksheets from wayground provide comprehensive printable practice problems and answer keys to help students master heat transfer calculations, specific heat capacity, and thermodynamic principles through engaging pdf exercises.

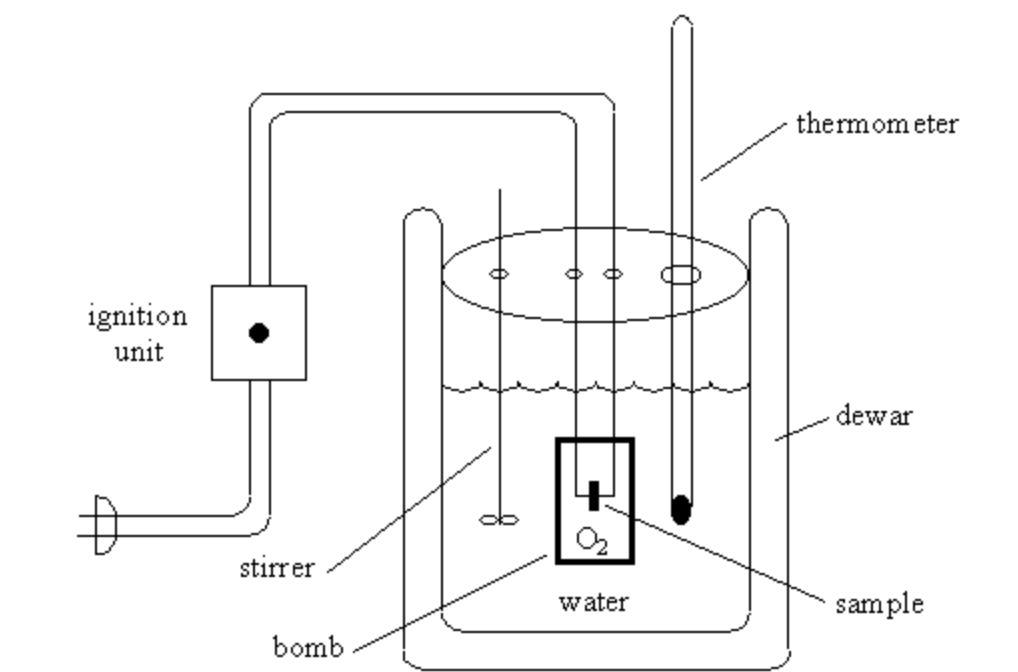
Calorimetry Unifyphysics When a hot object is placed in the calorimeter, heat energy is transferred from the object to the water and the water heats up. calorimeters can be used to find a substance’s specific heat capacity. What is a calorimeter? a calorimeter is just a container that is insulated. it holds a liquid that is usually water inside of it and you can have reactions happening inside of it as well. a thermometer will be able to read the temperature changes of the water. Grade 12 lesson plan on calorimetry, heat flow, and specific heat. includes activities, virtual experiments, and worksheets. physics and chemistry focused. Key words: calorimetry; calorimeter; coffee cup calorimeter; the principle of heat exchange; heat (q); algorithm; worked example. key concepts: "c alorimetry is used to measure the amount.

Calorimetry Grade 12 lesson plan on calorimetry, heat flow, and specific heat. includes activities, virtual experiments, and worksheets. physics and chemistry focused. Key words: calorimetry; calorimeter; coffee cup calorimeter; the principle of heat exchange; heat (q); algorithm; worked example. key concepts: "c alorimetry is used to measure the amount. The investigation could be improved by taking into account the temperature changes and thus energy transferred to the thermometer, tongs, and calorimeter. other improvements would be increasing the insulation of calorimeter and faster transfer from hot to cold water. A calorimeter is a device used to measure energy changes during a physical or chemical change what does a calorimeter typically consist of well insulated reaction chamber, tight fitting cover with holes, and some mechanism to stir the contents calorimetry equation q=mcΔt q > j 1 cal = 4.184 joules m > g c > j g∙°c t > °c. Practising these calorimetry calculations will help students become proficient with this concept. click below for a few resources that can be used in conjunction with a lesson to further demonstrate the concept of calorimetry. This is a power point on calorimetry, using the equation q = mct to solve problems, calorimeters, specific heat capacity, enthalpy of physical changes and calorimetry assumptions.

Calorimetry The investigation could be improved by taking into account the temperature changes and thus energy transferred to the thermometer, tongs, and calorimeter. other improvements would be increasing the insulation of calorimeter and faster transfer from hot to cold water. A calorimeter is a device used to measure energy changes during a physical or chemical change what does a calorimeter typically consist of well insulated reaction chamber, tight fitting cover with holes, and some mechanism to stir the contents calorimetry equation q=mcΔt q > j 1 cal = 4.184 joules m > g c > j g∙°c t > °c. Practising these calorimetry calculations will help students become proficient with this concept. click below for a few resources that can be used in conjunction with a lesson to further demonstrate the concept of calorimetry. This is a power point on calorimetry, using the equation q = mct to solve problems, calorimeters, specific heat capacity, enthalpy of physical changes and calorimetry assumptions.
Comments are closed.