Calorimetry Experiment Calculations

Calorimetry Experiment Calculations 1 Pdf Calorimetry Experiment Determine the specific heat capacity of a metal using a coffee cup calorimeter. heat is a form of energy that is transferred between objects with different temperatures. heat always flows from high temperature to low temperature. Explore calorimetry experiments in ib chemistry. learn how to calculate energy changes from temperature, mass, and specific heat capacity.
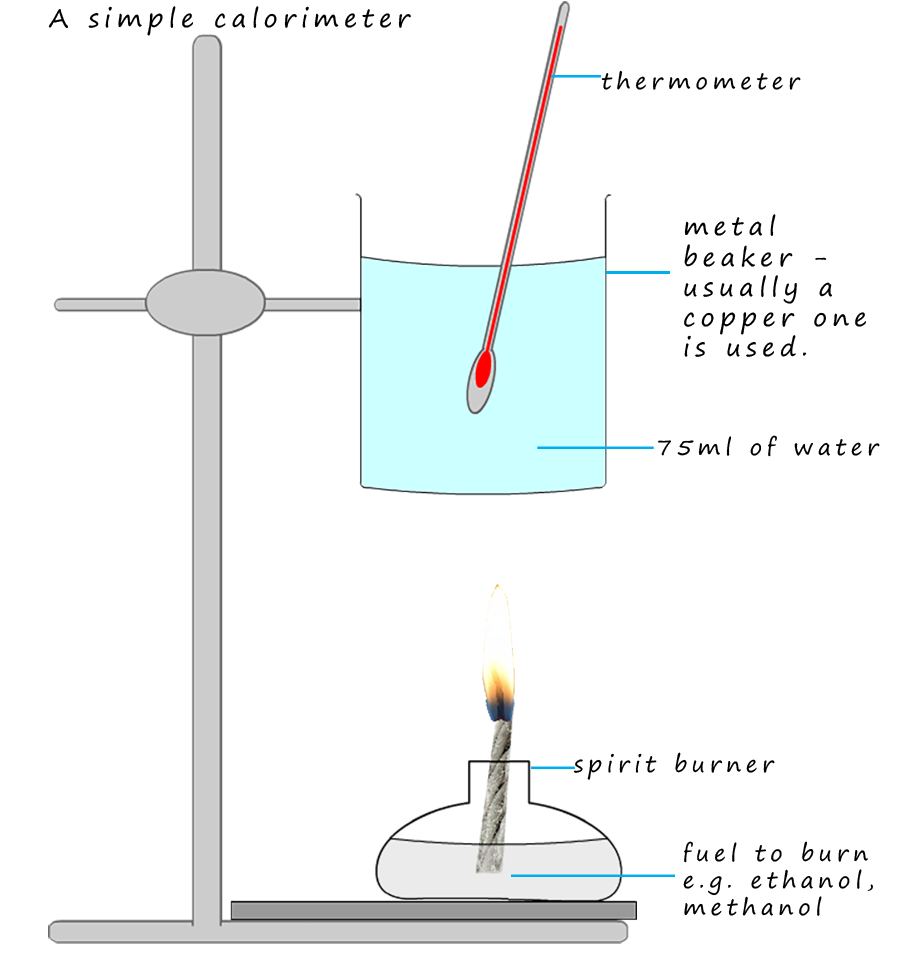
Calorimetry Experiment Alcohols At Andrew Gillan Blog Report: the laboratory report should include tables 1 4 (add titles to tables 3 and 4) with calculations, and the two graphs with titles, trend lines and tf identified. A bomb calorimeter is calibrated by combusting 1.00 g of ethanol, releasing 29.7 kj of energy, causing a temperature rise of 5.5°c in the calorimeter. after calibration, the calorimeter is then immediately used to determine the energy content of a biodiesel sample. Learn how calorimetry is used in thermochemistry to measure heat transferred during chemical reactions, phase changes, and specific heat experiments. Calorimetry is used to measure amounts of heat transferred to or from a substance. to do so, the heat is exchanged with a calibrated object (calorimeter). the temperature change measured by the calorimeter is used to derive the amount of heat transferred by the process under study.

Calorimetry Of Food Experiment At Lauren Hilson Blog Learn how calorimetry is used in thermochemistry to measure heat transferred during chemical reactions, phase changes, and specific heat experiments. Calorimetry is used to measure amounts of heat transferred to or from a substance. to do so, the heat is exchanged with a calibrated object (calorimeter). the temperature change measured by the calorimeter is used to derive the amount of heat transferred by the process under study. Before we practice calorimetry problems involving chemical reactions, consider a simpler example that illustrates the core idea behind calorimetry. suppose we initially have a high temperature substance, such as a hot piece of metal (m), and a low temperature substance, such as cool water (w). Experiment #8: calorimetry when a substance undergoes a change in temperature, the quantity (q) of heat lost or gained can be calculated using the mass (m), specific heat (s), and change in temperature (Δt = tfinal – tinitial):. In calorimetry, we measure the temperature change of a substance that surrounds a reaction. from this temperature change, we can calculate the energy change of the reaction. to then find the enthalpy change for the reaction, we would need to divide the energy change by the number of moles reacting. Add some cold water to the hot water contained in the calorimeter. place both probes inside the calorimeter and observe how the temperatures are changing until the equilibrium final temperature is reached.
Comments are closed.