Calorimetry
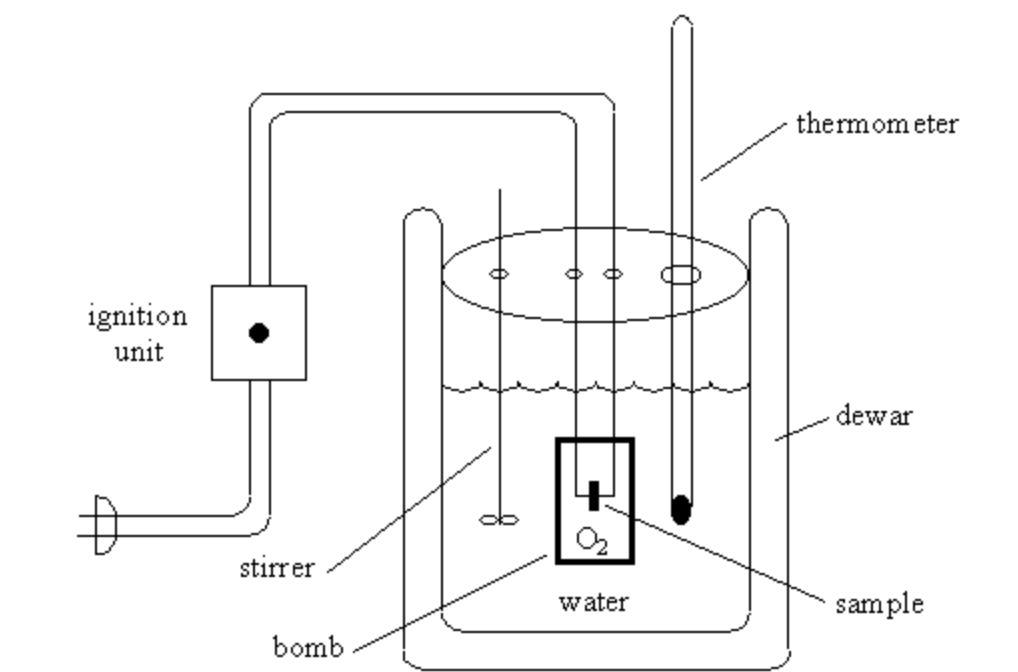
Calorimetry Chemtalk Calorimetry is the science of measuring heat transfer in chemical, physical or phase changes. learn about the history, types and equations of calorimetry, and how it is used in thermochemistry, biology and materials science. Learn what calorimetry is, how it works, and what types of calorimeters are used to measure heat exchange and enthalpy changes. find out how calorimetry is applied in chemistry, biology, and daily life with examples and formulas.

Calorimetry Chemtalk Calorimetry refers to a set of techniques used to measure energy changes in chemical processes using devices called calorimeters. for these measurements to be meaningful, the temperature change observed must be related to the amount of heat absorbed or released by the system. Calorimetry is used to measure the amount of thermal energy transferred in a chemical or physical process. this requires careful measurement of the temperature change that occurs during the process and the masses of the system and surroundings. Learn how to measure the amount of heat involved in a physical or chemical reaction using calorimeters and the calorimetry equation. explore different types of calorimeters, endothermic and exothermic reactions, and practice problems with solutions. What is calorimetry? calorimetry is defined as the process of measuring the heat capacity, specific heat, or the heat absorbed or released during a chemical or physical change. this science is founded on the basic principles of heat measurement and energy transfer, mainly the conservation of energy.
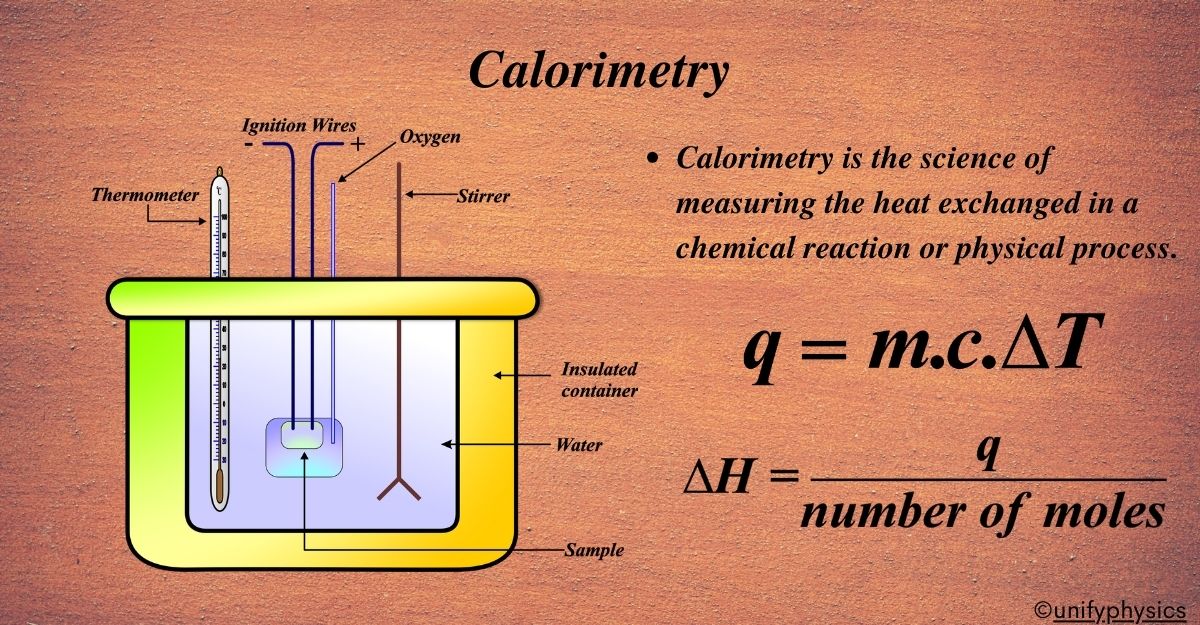
Calorimetry Unifyphysics Learn how to measure the amount of heat involved in a physical or chemical reaction using calorimeters and the calorimetry equation. explore different types of calorimeters, endothermic and exothermic reactions, and practice problems with solutions. What is calorimetry? calorimetry is defined as the process of measuring the heat capacity, specific heat, or the heat absorbed or released during a chemical or physical change. this science is founded on the basic principles of heat measurement and energy transfer, mainly the conservation of energy. One technique we can use to measure the amount of heat involved in a chemical or physical process is known as calorimetry. calorimetry is used to measure amounts of heat transferred to or from a substance. to do so, the heat is exchanged with a calibrated object (calorimeter). Calorimetry is a collection of experimental techniques used to measure energy changes and heat flows associated with chemical reactions and physical changes. the term "calorimetry" comes from the greek words for heat ("calor") and measure ("meter"). Learn how to measure heat capacity and specific heat of substances using calorimetry. find definitions, examples, equations, and data tables for common substances. Calorimetry is the technique chemists and students use to measure how much heat is released or absorbed by a system. by tracking temperature changes in the surroundings, we can quantify energy transfer and better understand reaction energetics, phase changes, and heat capacity.
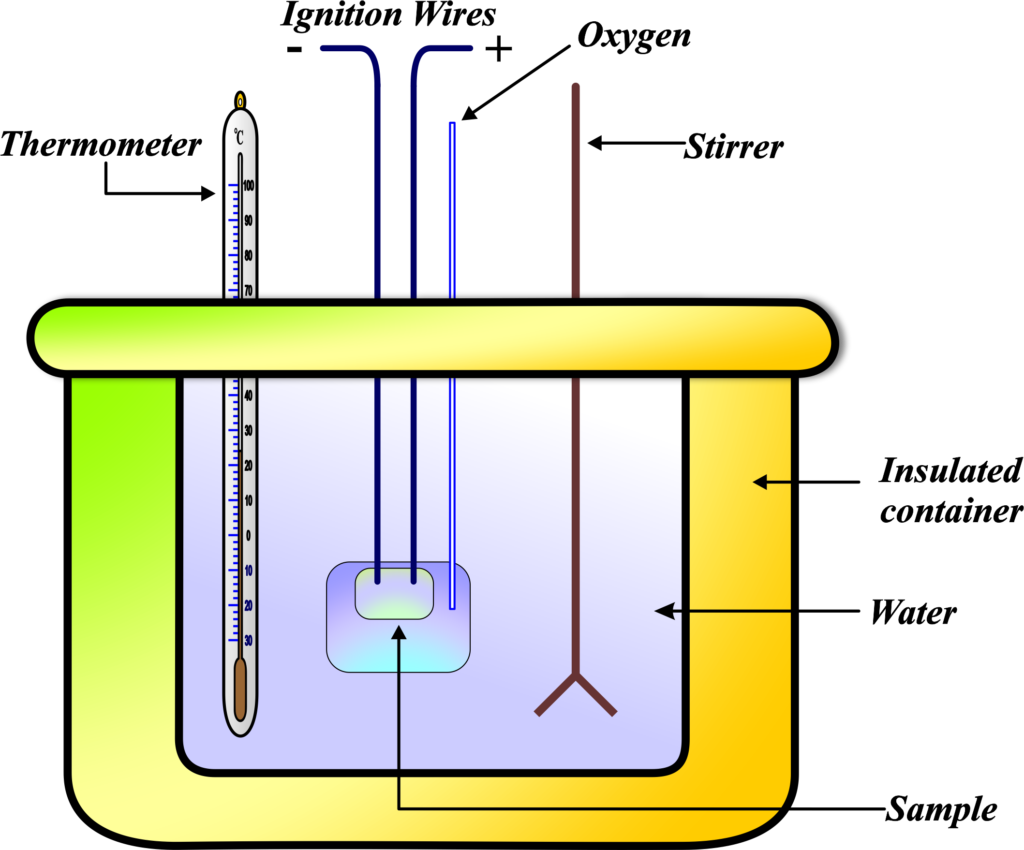
Calorimetry Unifyphysics One technique we can use to measure the amount of heat involved in a chemical or physical process is known as calorimetry. calorimetry is used to measure amounts of heat transferred to or from a substance. to do so, the heat is exchanged with a calibrated object (calorimeter). Calorimetry is a collection of experimental techniques used to measure energy changes and heat flows associated with chemical reactions and physical changes. the term "calorimetry" comes from the greek words for heat ("calor") and measure ("meter"). Learn how to measure heat capacity and specific heat of substances using calorimetry. find definitions, examples, equations, and data tables for common substances. Calorimetry is the technique chemists and students use to measure how much heat is released or absorbed by a system. by tracking temperature changes in the surroundings, we can quantify energy transfer and better understand reaction energetics, phase changes, and heat capacity.

Calorimetry Learn how to measure heat capacity and specific heat of substances using calorimetry. find definitions, examples, equations, and data tables for common substances. Calorimetry is the technique chemists and students use to measure how much heat is released or absorbed by a system. by tracking temperature changes in the surroundings, we can quantify energy transfer and better understand reaction energetics, phase changes, and heat capacity.
Comments are closed.