Bs En Iso 9626 2016 Stainless Steel Needle Tubing For The Manufacture

Bs En Iso 9626 2016 Stainless Steel Needle Tubing For The Manufacture Iso 9626:2016 applies to rigid stainless steel needle tubing suitable for use in the manufacture of hypodermic needles and other medical devices primarily for human use. it provides requirements and test methods for the tubes manufactured for needles as component used in medical devices. Rstellung von medizinprodukten anforderungen und prüfverfahren (iso 9626:2016) this european standard was approved by cen on 12 june 2016. cen members are bound to comply with the cen cenelec.

Bs En Iso 9626 2016 Stainless Steel Needle Tubing For The Manufacture This international standard provides requirements and test methods for the tubes manufactured for needles as component used in medical devices. additional performance testing on the tube aspect may be required when the component is incorporated in the ready to use device. Iso 9626:2016 applies to rigid stainless steel needle tubing suitable for use in the manufacture of hypodermic needles and other medical devices primarily for human use. it provides requirements and test methods for the tubes manufactured for needles as component used in medical devices. Stainless steel needle tubing for the manufacture of medical devices — requirements and test methods. Stainless steel needle tubing for the manufacture of medical devices. requirements and test methods. bs en iso 9626:2016 is maintained by ch 84. this standard is available from the following sources:.

Din En Iso 9626 Stainless steel needle tubing for the manufacture of medical devices — requirements and test methods. Stainless steel needle tubing for the manufacture of medical devices. requirements and test methods. bs en iso 9626:2016 is maintained by ch 84. this standard is available from the following sources:. Iso 9626:2016 applies to rigid stainless steel needle tubing suitable for use in the manufacture of hypodermic needles and other medical devices primarily for human use. it provides requirements and test methods for the tubes manufactured for needles as component used in medical devices. Bs en iso 9626:2016 applies to rigid stainless steel needle tubing suitable for use in the manufacture of hypodermic needles and other medical devices primarily for human use. this international standard provides requirements and test methods for the tubes manufactured for needles as component used in medical devices. This international standard applies to rigid stainless steel needle tubing suitable for use in the manufacture of hypodermic needles and other medical devices primarily for human use. Iso 9626:2016 applies to rigid stainless steel needle tubing suitable for use in the manufacture of hypodermic needles and other medical devices primarily for human use. it provides requirements and test methods for the tubes manufactured for needles as component used in medical devices.

Din En Iso 9626 Iso 9626:2016 applies to rigid stainless steel needle tubing suitable for use in the manufacture of hypodermic needles and other medical devices primarily for human use. it provides requirements and test methods for the tubes manufactured for needles as component used in medical devices. Bs en iso 9626:2016 applies to rigid stainless steel needle tubing suitable for use in the manufacture of hypodermic needles and other medical devices primarily for human use. this international standard provides requirements and test methods for the tubes manufactured for needles as component used in medical devices. This international standard applies to rigid stainless steel needle tubing suitable for use in the manufacture of hypodermic needles and other medical devices primarily for human use. Iso 9626:2016 applies to rigid stainless steel needle tubing suitable for use in the manufacture of hypodermic needles and other medical devices primarily for human use. it provides requirements and test methods for the tubes manufactured for needles as component used in medical devices.
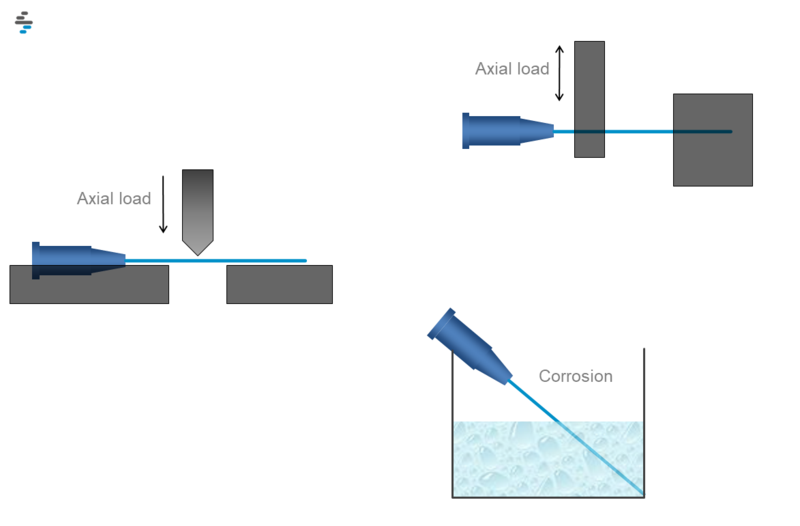
Standard This international standard applies to rigid stainless steel needle tubing suitable for use in the manufacture of hypodermic needles and other medical devices primarily for human use. Iso 9626:2016 applies to rigid stainless steel needle tubing suitable for use in the manufacture of hypodermic needles and other medical devices primarily for human use. it provides requirements and test methods for the tubes manufactured for needles as component used in medical devices.
Comments are closed.