Breakthrough Device Designation Timeline

Breakthrough Device Designation Mastertrial Learn about the breakthrough devices program benefits, criteria and how to request a designation. A breakthrough device designation (bdd) request is a stand alone request to fda; it cannot be combined with any other requests such as a pre submission meeting. fda will issue a grant or denial decision for each bdd request within 60 calendar days of receipt.

Breakthrough Device Designation Timeline This document provides guidance for a process that accommodates the needs of these devices and provides considerations for the clinical or performance evaluation of breakthrough medical devices (btmd) and breakthrough in vitro diagnostic medical devices (btivd). For medical device manufacturers, understanding whether the fda breakthrough device designation applies to your technology can significantly influence regulatory strategy, development timelines and downstream commercialization planning. Proxima's director of regulatory affairs and quality assurance, ellie reynolds, walks through the complete bdd submission timeline, from initial application to fda decision, so you know exactly what to expect during this expedited approval process. Fda's breakthrough devices program expedites review by 6 12 months for qualifying medical devices. learn eligibility requirements, benefits & application strategy.

Breakthrough Device Designation Granted Scinvivo Proxima's director of regulatory affairs and quality assurance, ellie reynolds, walks through the complete bdd submission timeline, from initial application to fda decision, so you know exactly what to expect during this expedited approval process. Fda's breakthrough devices program expedites review by 6 12 months for qualifying medical devices. learn eligibility requirements, benefits & application strategy. According to the fda, as of june 30, 2025, 1,176 devices have been granted breakthrough device designation. this number reflects a significant pipeline of novel technologies moving through the regulatory process. Learn how fda breakthrough device designation accelerates medical innovation. explore benefits, eligibility, and expert guidance for your device approval. As of june 30, 2023, cdrh has granted breakthrough device designation to 831 devices and granted marketing authorization to 77 devices with breakthrough device designation. these. The breakthrough devices program is a voluntary program for certain medical devices and device led combination products that provide for more effective treatment or diagnosis of life threatening or irreversibly debilitating diseases or conditions.
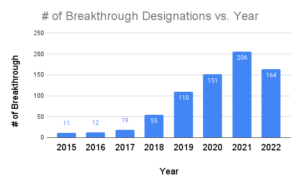
What Is An Fda Breakthrough Device Designation According to the fda, as of june 30, 2025, 1,176 devices have been granted breakthrough device designation. this number reflects a significant pipeline of novel technologies moving through the regulatory process. Learn how fda breakthrough device designation accelerates medical innovation. explore benefits, eligibility, and expert guidance for your device approval. As of june 30, 2023, cdrh has granted breakthrough device designation to 831 devices and granted marketing authorization to 77 devices with breakthrough device designation. these. The breakthrough devices program is a voluntary program for certain medical devices and device led combination products that provide for more effective treatment or diagnosis of life threatening or irreversibly debilitating diseases or conditions.
Comments are closed.