Breakthrough Device Designation

Breakthrough Device Designation Mastertrial Learn about the breakthrough devices program benefits, criteria and how to request a designation. Learn how to apply for fda's breakthrough devices program, which provides faster and more interactive review for medical devices with significant innovation or unmet need. find out the criteria, data requirements, submission mechanisms, and resources for this program.

Breakthrough Device Designation Granted Scinvivo The fda breakthrough device designation program faces a critical juncture. while it has facilitated some innovative device approvals and provides investor credibility, current evidence suggests diminishing returns, particularly for devices suitable for 510 (k) clearance. What does fda breakthrough device designation mean? for medical device manufacturers, understanding whether the fda breakthrough device designation applies to your technology can significantly influence regulatory strategy, development timelines and downstream commercialization planning. Fda's breakthrough devices program expedites review by 6 12 months for qualifying medical devices. learn eligibility requirements, benefits & application strategy. Learn how fda breakthrough device designation accelerates medical innovation. explore benefits, eligibility, and expert guidance for your device approval.
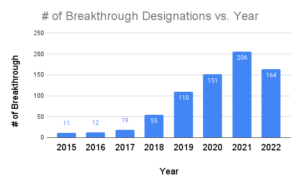
What Is An Fda Breakthrough Device Designation Fda's breakthrough devices program expedites review by 6 12 months for qualifying medical devices. learn eligibility requirements, benefits & application strategy. Learn how fda breakthrough device designation accelerates medical innovation. explore benefits, eligibility, and expert guidance for your device approval. The fda gives the designation to devices that may provide more effective treatment or diagnosis of life threatening or debilitating conditions. in its 2025 fiscal year, which began in october, the agency had granted 136 breakthrough designations as of june 30. The breakthrough devices program is intended to expedite the development of, and prioritise the us food and drug administration’s (fda’s) review of, certain medical devices designated as. Learn how to fast track your medical device through the fda with the breakthrough device designation, a special status for innovative technologies that can improve patient outcomes. find out the benefits, criteria, and application process for this program. Breakthrough device designation (bdd) is a free, fast fda program that signals serious unmet need and potential meaningful improvement. for ai ml samd teams, it de risks pivotal study strategy through earlier, higher touch fda engagement (including tap), faster decision cycles, and clearer evidence expectations.

What Is The Fda Breakthrough Device Designation The fda gives the designation to devices that may provide more effective treatment or diagnosis of life threatening or debilitating conditions. in its 2025 fiscal year, which began in october, the agency had granted 136 breakthrough designations as of june 30. The breakthrough devices program is intended to expedite the development of, and prioritise the us food and drug administration’s (fda’s) review of, certain medical devices designated as. Learn how to fast track your medical device through the fda with the breakthrough device designation, a special status for innovative technologies that can improve patient outcomes. find out the benefits, criteria, and application process for this program. Breakthrough device designation (bdd) is a free, fast fda program that signals serious unmet need and potential meaningful improvement. for ai ml samd teams, it de risks pivotal study strategy through earlier, higher touch fda engagement (including tap), faster decision cycles, and clearer evidence expectations.
Comments are closed.