Bohrs Atomic Model Labeled

Bohr S Atomic Model Postulates Scienly Bohr model of all elements is mentioned in the chart below. details about this periodic table: access detailed info on all elements: atomic mass, electron configurations, charges, and more. view rotating bohr models for all 118 elements. get a free hd image of the periodic table. Learn all about the bohr model of atomic structure, with many clear examples, diagrams of atoms, history and comparisons to other atomic models.
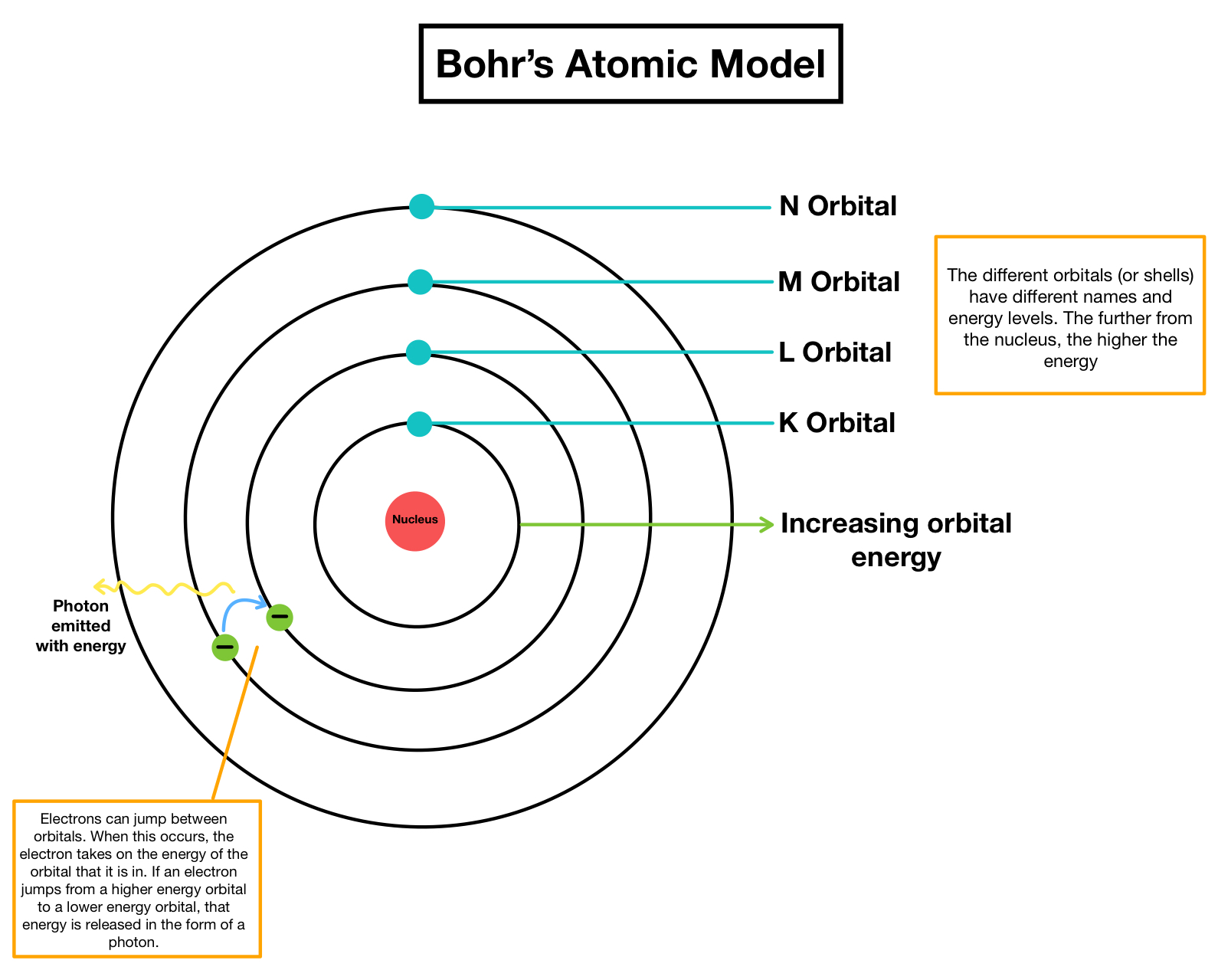
Bohr S Atomic Model Overview Importance Expii Bohr model of the atom in the bohr model of the atom, electrons travel in defined circular orbits around the nucleus. the orbits are labeled by an integer, the quantum number n. electrons can jump from one orbit to another by emitting or absorbing energy. Learn about the bohr model of the atom. see the main points of the model, how to calculate absorbed or emitted energy, and why the model is important. Bohr atomic model described & explained with examples, diagrams, & problems. what does it represent. learn its history, main postulates, theory, & formula. Bohr’s model of an atom explains that in an atom all the electrons revolve around the positively charged center called the nucleus. according to this model, the electron revolving near the nucleus has less energy, whereas the electron revolving away from the nucleus has higher energy.

Define The Concept Of The Bohrs Atomic Model Bohr atomic model described & explained with examples, diagrams, & problems. what does it represent. learn its history, main postulates, theory, & formula. Bohr’s model of an atom explains that in an atom all the electrons revolve around the positively charged center called the nucleus. according to this model, the electron revolving near the nucleus has less energy, whereas the electron revolving away from the nucleus has higher energy. Bohr model of the atom in this section, we describe the structure and behaviour of the simplest type of atom consisting of a nucleus orbited by a single electron only. it addresses the basic question: how do electrons remain in stable orbits around the atomic nucleus?. Discover bohr’s atomic model with easy postulates, energy levels, diagrams, and real life examples. master concepts for neet, jee, and board exams. The electron shells are labeled k, l, m, n, o, p, and q; or 1, 2, 3, 4, 5, 6, and 7; going from innermost shell outwards. electrons in outer shells have higher average energy and travel farther from the nucleus than those in inner shells. The bohr model (named after danish physicist niels bohr) of an atom has a small, positively charged central nucleus and electrons orbiting in at specific fixed distances from the nucleus.
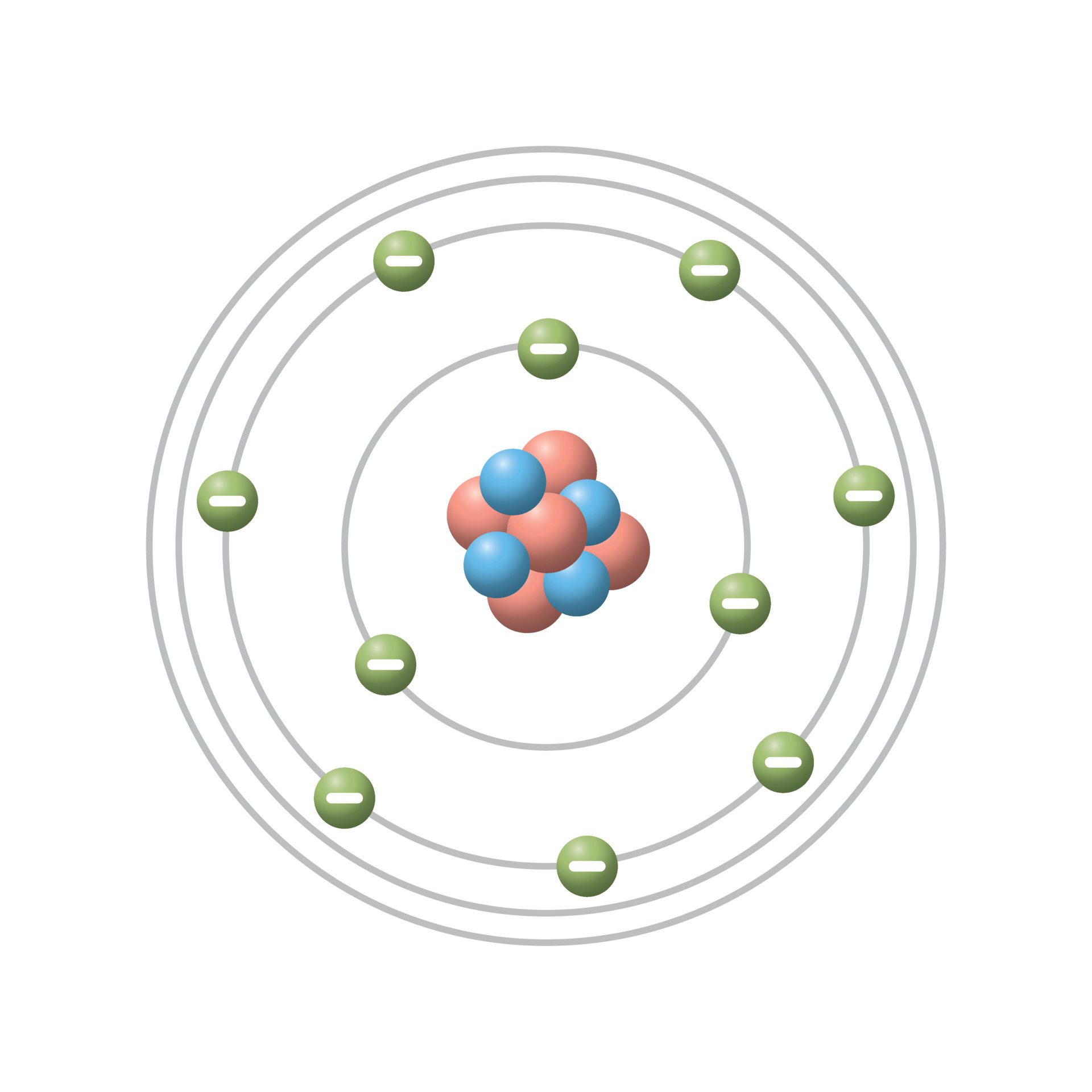
Bohr S Model Of Atom Tolfarea Bohr model of the atom in this section, we describe the structure and behaviour of the simplest type of atom consisting of a nucleus orbited by a single electron only. it addresses the basic question: how do electrons remain in stable orbits around the atomic nucleus?. Discover bohr’s atomic model with easy postulates, energy levels, diagrams, and real life examples. master concepts for neet, jee, and board exams. The electron shells are labeled k, l, m, n, o, p, and q; or 1, 2, 3, 4, 5, 6, and 7; going from innermost shell outwards. electrons in outer shells have higher average energy and travel farther from the nucleus than those in inner shells. The bohr model (named after danish physicist niels bohr) of an atom has a small, positively charged central nucleus and electrons orbiting in at specific fixed distances from the nucleus.
Comments are closed.