Bohrs Atomic Model Golfshanghai

Bohrs Atomic Model Atomic Spectrum Auto Saved Pdf Atomic Just as bohr imagined that the atoms were little planets revolving around the nucleus as though it were a sun, other scientists tried to extend that idea by imagining that the little planets, just like real planets, had spin. It consists of a small, dense atomic nucleus surrounded by orbiting electrons. it is analogous to the structure of the solar system, but with attraction provided by electrostatic force rather than gravity, and with the electron energies quantized (assuming only discrete values).

Bohr S Atomic Model The bohr model of the atom was proposed by the danish physicist niels bohr in 1913. the bohr model of the atom, a radical departure from earlier, classical descriptions, was the first to incorporate quantum mechanics. Bohr’s model of an atom explains that in an atom all the electrons revolve around the positively charged center called the nucleus. according to this model, the electron revolving near the nucleus has less energy, whereas the electron revolving away from the nucleus has higher energy. This page explores the analogy of climbing a ladder to explain potential energy and niels bohr's 1915 atomic model, where electrons occupy fixed energy states around the nucleus. Bohr model of the atom in this section, we describe the structure and behaviour of the simplest type of atom consisting of a nucleus orbited by a single electron only. it addresses the basic question: how do electrons remain in stable orbits around the atomic nucleus?.

Bohr S Atomic Model Energy Transitions And Electron Orbits This page explores the analogy of climbing a ladder to explain potential energy and niels bohr's 1915 atomic model, where electrons occupy fixed energy states around the nucleus. Bohr model of the atom in this section, we describe the structure and behaviour of the simplest type of atom consisting of a nucleus orbited by a single electron only. it addresses the basic question: how do electrons remain in stable orbits around the atomic nucleus?. The bohr model of the atom was proposed by niels bohr in 1913 as an expansion on and correction of the rutherford model. his model depicted atoms as having negatively charged electrons which orbited a small, positively charged nuclei containing most of the atom's mass, as rutherford had done. Try out different models by shooting light at the atom. check how the prediction of the model matches the experimental results. The bohr model represents the structure of an atom developed by danish physicist niels bohr in 1913. according to this model, the atomic structure is similar to that of the solar system. In bohr’s atomic model, electrons are in a given orbit. this model is also called a planetary model because electrons orbiting around an atomic nucleus resemble a planet orbiting the sun.
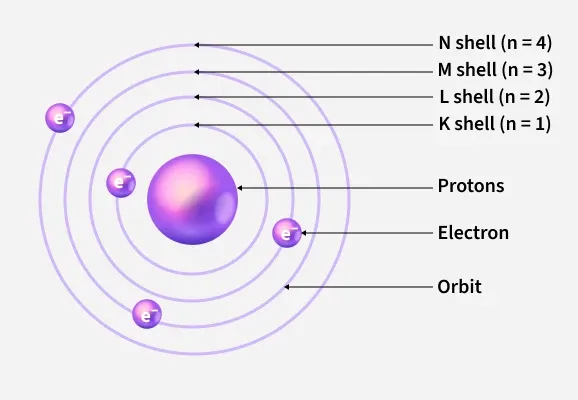
Bohrs Atomic Model The bohr model of the atom was proposed by niels bohr in 1913 as an expansion on and correction of the rutherford model. his model depicted atoms as having negatively charged electrons which orbited a small, positively charged nuclei containing most of the atom's mass, as rutherford had done. Try out different models by shooting light at the atom. check how the prediction of the model matches the experimental results. The bohr model represents the structure of an atom developed by danish physicist niels bohr in 1913. according to this model, the atomic structure is similar to that of the solar system. In bohr’s atomic model, electrons are in a given orbit. this model is also called a planetary model because electrons orbiting around an atomic nucleus resemble a planet orbiting the sun.

Bohr S Atomic Model Postulates Scienly The bohr model represents the structure of an atom developed by danish physicist niels bohr in 1913. according to this model, the atomic structure is similar to that of the solar system. In bohr’s atomic model, electrons are in a given orbit. this model is also called a planetary model because electrons orbiting around an atomic nucleus resemble a planet orbiting the sun.
Comments are closed.