Bohrs Atomic Model 2

Bohrs Atomic Model Atomic Spectrum Auto Saved Download Free Pdf In atomic physics, the bohr model or rutherford–bohr model is an obsolete model of the atom that incorporated some early quantum concepts. In this model, the electrons orbit the nucleus in circular orbits, accounting for the series of discrete wavelengths in the h2 emission spectrum. bohr's model of the hydrogen atom was the first to incorporate quantum theory, and the key idea of his model was that electrons occupy discrete orbitals.

Bohr S Atomic Model The bohr model of the atom was proposed by the danish physicist niels bohr in 1913. the bohr model of the atom, a radical departure from earlier, classical descriptions, was the first to incorporate quantum mechanics. Bohr's model is an atomic model proposed by danish physicist niels bohr in 1913. in an atom, the electrons revolve around the nucleus in definite energy levels called orbits shells. this model provides a basic understanding of the concept of the atom and its constituents. Learn about the bohr model of the atom. see the main points of the model, how to calculate absorbed or emitted energy, and why the model is important. Bohr’s model of the hydrogen atom, proposed by niels bohr in 1913, was the first quantum model that correctly explained the hydrogen emission spectrum. bohr’s model combines the classical mechanics of planetary motion with the quantum concept of photons.

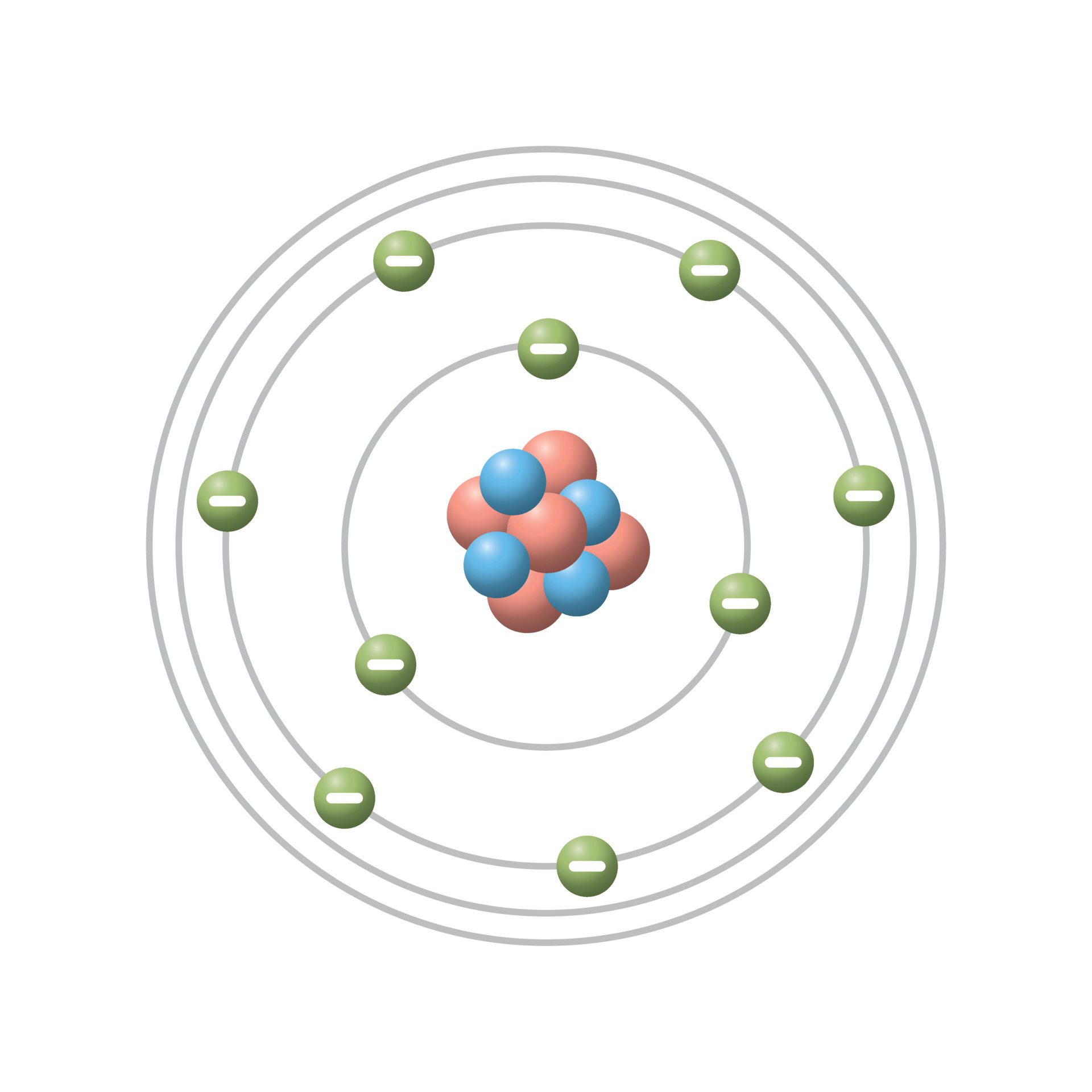
Bohr S Atomic Model Postulates Scienly Learn about the bohr model of the atom. see the main points of the model, how to calculate absorbed or emitted energy, and why the model is important. Bohr’s model of the hydrogen atom, proposed by niels bohr in 1913, was the first quantum model that correctly explained the hydrogen emission spectrum. bohr’s model combines the classical mechanics of planetary motion with the quantum concept of photons. Bohr model of the atom in this section, we describe the structure and behaviour of the simplest type of atom consisting of a nucleus orbited by a single electron only. it addresses the basic question: how do electrons remain in stable orbits around the atomic nucleus?. In 1913, niels bohr attempted to resolve the atomic paradox by ignoring classical electromagnetism’s prediction that the orbiting electron in hydrogen would continuously emit light. Bohr atomic model described & explained with examples, diagrams, & problems. what does it represent. learn its history, main postulates, theory, & formula. When electrons jump from one orbit to another, energy is gained or lost in the form of light! hypothesis: wavelengths of electrons are related to energy and only orbits with integer wavelengths are allowed. a drawing of the bohr model of electron orbits in the hydrogen atom is shown.

Bohr S Atomic Model Postulates Scienly Bohr model of the atom in this section, we describe the structure and behaviour of the simplest type of atom consisting of a nucleus orbited by a single electron only. it addresses the basic question: how do electrons remain in stable orbits around the atomic nucleus?. In 1913, niels bohr attempted to resolve the atomic paradox by ignoring classical electromagnetism’s prediction that the orbiting electron in hydrogen would continuously emit light. Bohr atomic model described & explained with examples, diagrams, & problems. what does it represent. learn its history, main postulates, theory, & formula. When electrons jump from one orbit to another, energy is gained or lost in the form of light! hypothesis: wavelengths of electrons are related to energy and only orbits with integer wavelengths are allowed. a drawing of the bohr model of electron orbits in the hydrogen atom is shown.

Define The Concept Of The Bohrs Atomic Model Bohr atomic model described & explained with examples, diagrams, & problems. what does it represent. learn its history, main postulates, theory, & formula. When electrons jump from one orbit to another, energy is gained or lost in the form of light! hypothesis: wavelengths of electrons are related to energy and only orbits with integer wavelengths are allowed. a drawing of the bohr model of electron orbits in the hydrogen atom is shown.
Bohr S Model Of Atom Tolfarea
Comments are closed.