Bohrs Atomic Model

Bohr S Atomic Model In atomic physics, the bohr model or rutherford–bohr model is an obsolete model of the atom that incorporated some early quantum concepts. The bohr model is a description of the structure of atoms proposed in 1913 by the danish physicist niels bohr. the bohr model of the atom, a radical departure from earlier, classical descriptions, was the first to incorporate quantum theory and was the predecessor of wholly quantum mechanical models.

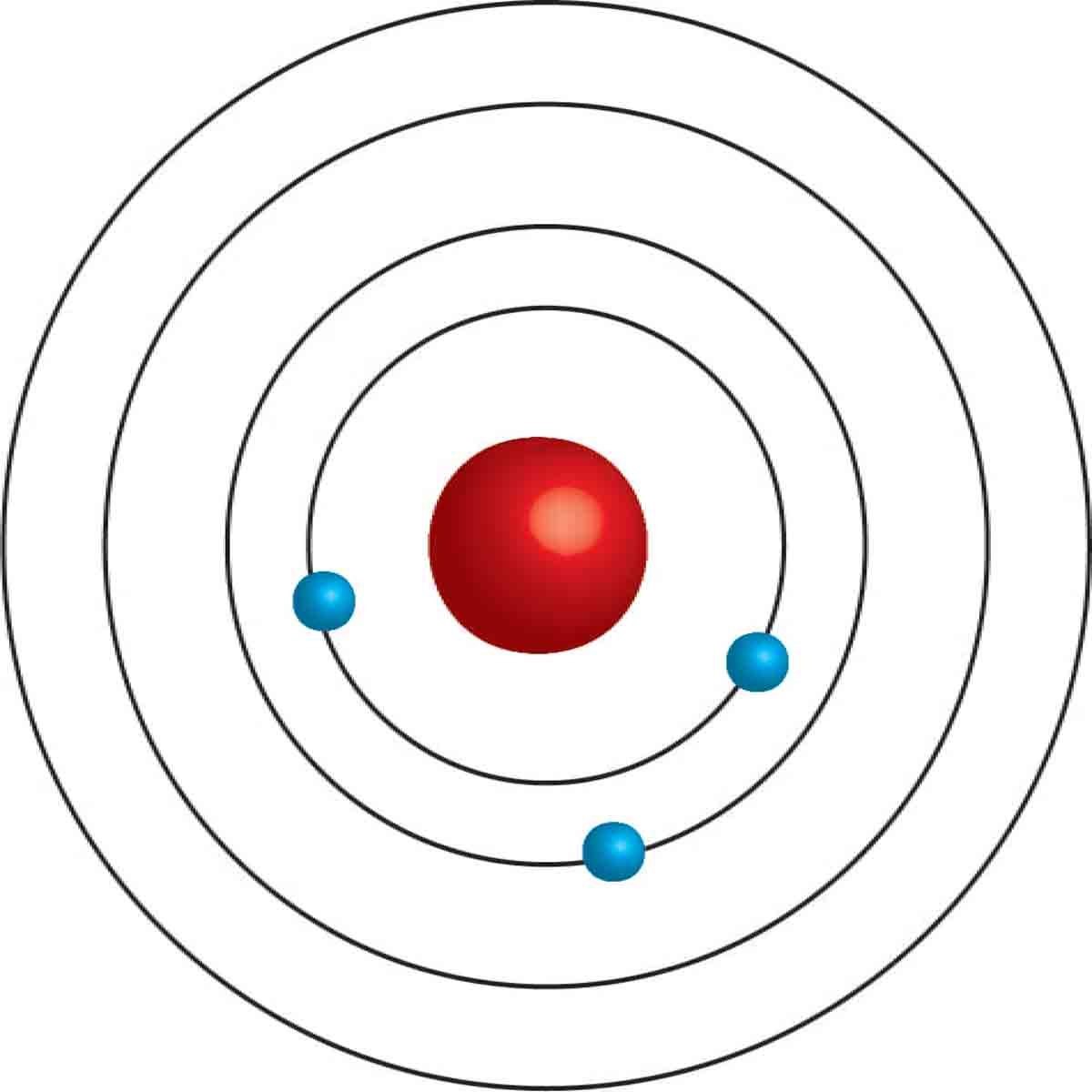
Bohrs Atomic Model Kwbooy Bohr's model is an atomic model proposed by danish physicist niels bohr in 1913. in an atom, the electrons revolve around the nucleus in definite energy levels called orbits shells. this model provides a basic understanding of the concept of the atom and its constituents. Learn about the bohr model, a cake or planetary model of the atom based on quantum theory. find out its key points, applications, problems, and improvements. Learn about the bohr model of the atom, which explains the spectral lines of hydrogen and hydrogen like atoms. find out the equations for radius, energy, and angular momentum of the electron orbits, and the limitations of the model. What is the bohr model? the bohr model is a model of an atom. the model was proposed by physicist niels bohr in 1913. in this model, the electrons travel around the nucleus of an atom in distinct circular orbits, or shells. the model is also referred to as the planetary model of an atom.

Bohr S Atomic Model Postulates Scienly Learn about the bohr model of the atom, which explains the spectral lines of hydrogen and hydrogen like atoms. find out the equations for radius, energy, and angular momentum of the electron orbits, and the limitations of the model. What is the bohr model? the bohr model is a model of an atom. the model was proposed by physicist niels bohr in 1913. in this model, the electrons travel around the nucleus of an atom in distinct circular orbits, or shells. the model is also referred to as the planetary model of an atom. Bohr's model of the hydrogen atom was the first to incorporate quantum theory, and the key idea of his model was that electrons occupy discrete orbitals. the bohr model of the atom was proposed by niels bohr in 1913 as an expansion on and correction of the rutherford model. In 1913, niels bohr attempted to resolve the atomic paradox by ignoring classical electromagnetism’s prediction that the orbiting electron in hydrogen would continuously emit light. What is bohr’s model in chemistry? a bohr’s model refers to an atomic model which proposes that electrons revolve around the nucleus in fixed, quantized orbits with specific energy levels. this model, introduced by niels bohr, links classical ideas and emerging quantum theory. Learn about the bohr model of an atom, which explains the structure and energy levels of electrons in an atom. find out the postulates, limitations and applications of this model, and watch related videos and faqs.
/Bohr-58e690203df78c51620ff02e.jpg)
Bohrs Atomic Model Golfshanghai Bohr's model of the hydrogen atom was the first to incorporate quantum theory, and the key idea of his model was that electrons occupy discrete orbitals. the bohr model of the atom was proposed by niels bohr in 1913 as an expansion on and correction of the rutherford model. In 1913, niels bohr attempted to resolve the atomic paradox by ignoring classical electromagnetism’s prediction that the orbiting electron in hydrogen would continuously emit light. What is bohr’s model in chemistry? a bohr’s model refers to an atomic model which proposes that electrons revolve around the nucleus in fixed, quantized orbits with specific energy levels. this model, introduced by niels bohr, links classical ideas and emerging quantum theory. Learn about the bohr model of an atom, which explains the structure and energy levels of electrons in an atom. find out the postulates, limitations and applications of this model, and watch related videos and faqs.

Bohrs Atomic Model And Its Significance Chemistry 11 2025 What is bohr’s model in chemistry? a bohr’s model refers to an atomic model which proposes that electrons revolve around the nucleus in fixed, quantized orbits with specific energy levels. this model, introduced by niels bohr, links classical ideas and emerging quantum theory. Learn about the bohr model of an atom, which explains the structure and energy levels of electrons in an atom. find out the postulates, limitations and applications of this model, and watch related videos and faqs.
The Basics Of Bohr S Model
Comments are closed.