Bohr Model Of Hydrogen Atom Unifyphysics
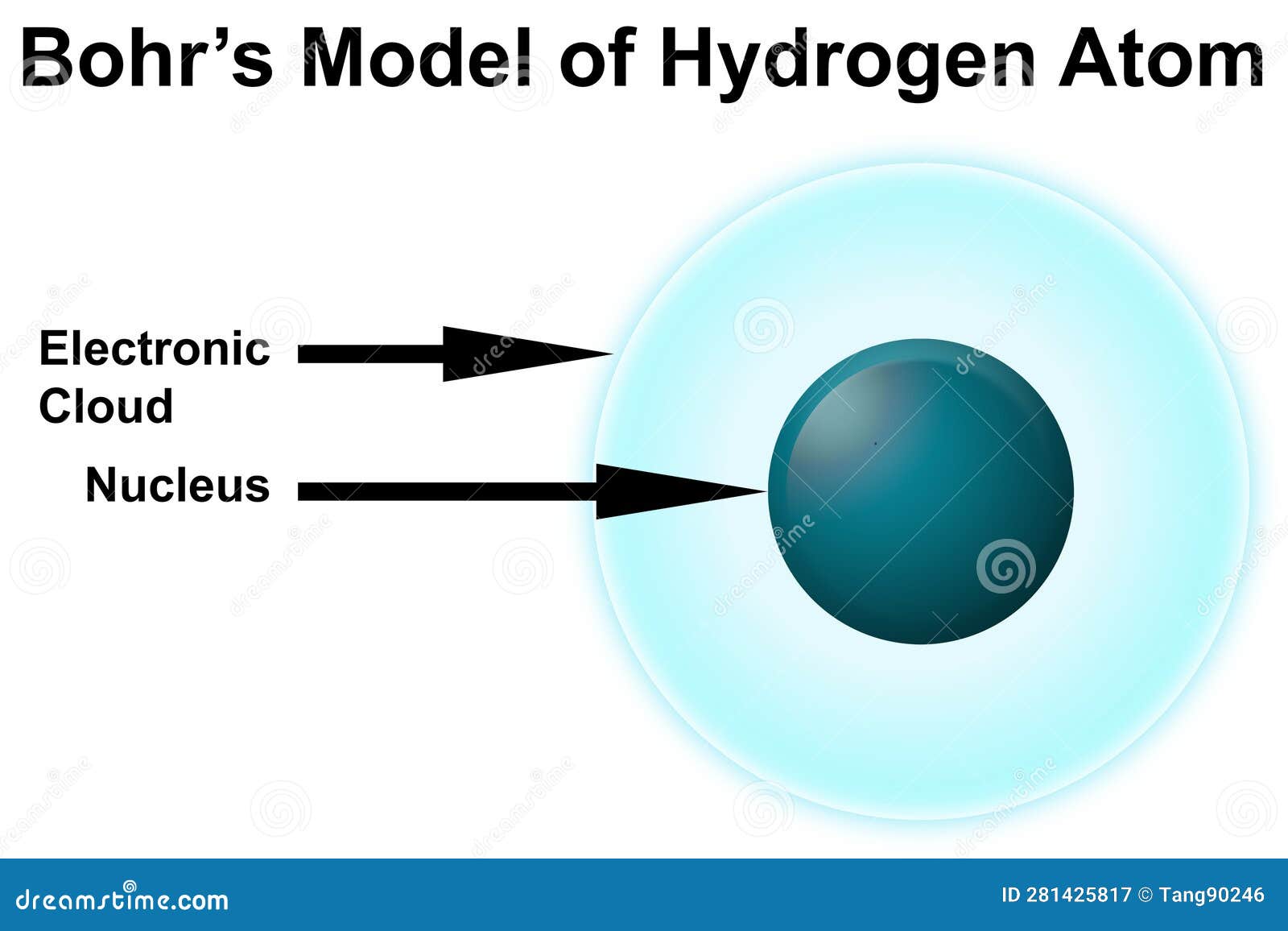
Bohr Model Of Hydrogen Atom Royalty Free Stock Photography The bohr model, often referred to as the planetary model of the atom, is a concept that simplifies the complex nature of atomic structures. it’s particularly focused on the hydrogen atom, which is the simplest atom with only one electron. Bohr’s model of the hydrogen atom, proposed by niels bohr in 1913, was the first quantum model that correctly explained the hydrogen emission spectrum. bohr’s model combines the classical mechanics of planetary motion with the quantum concept of photons.
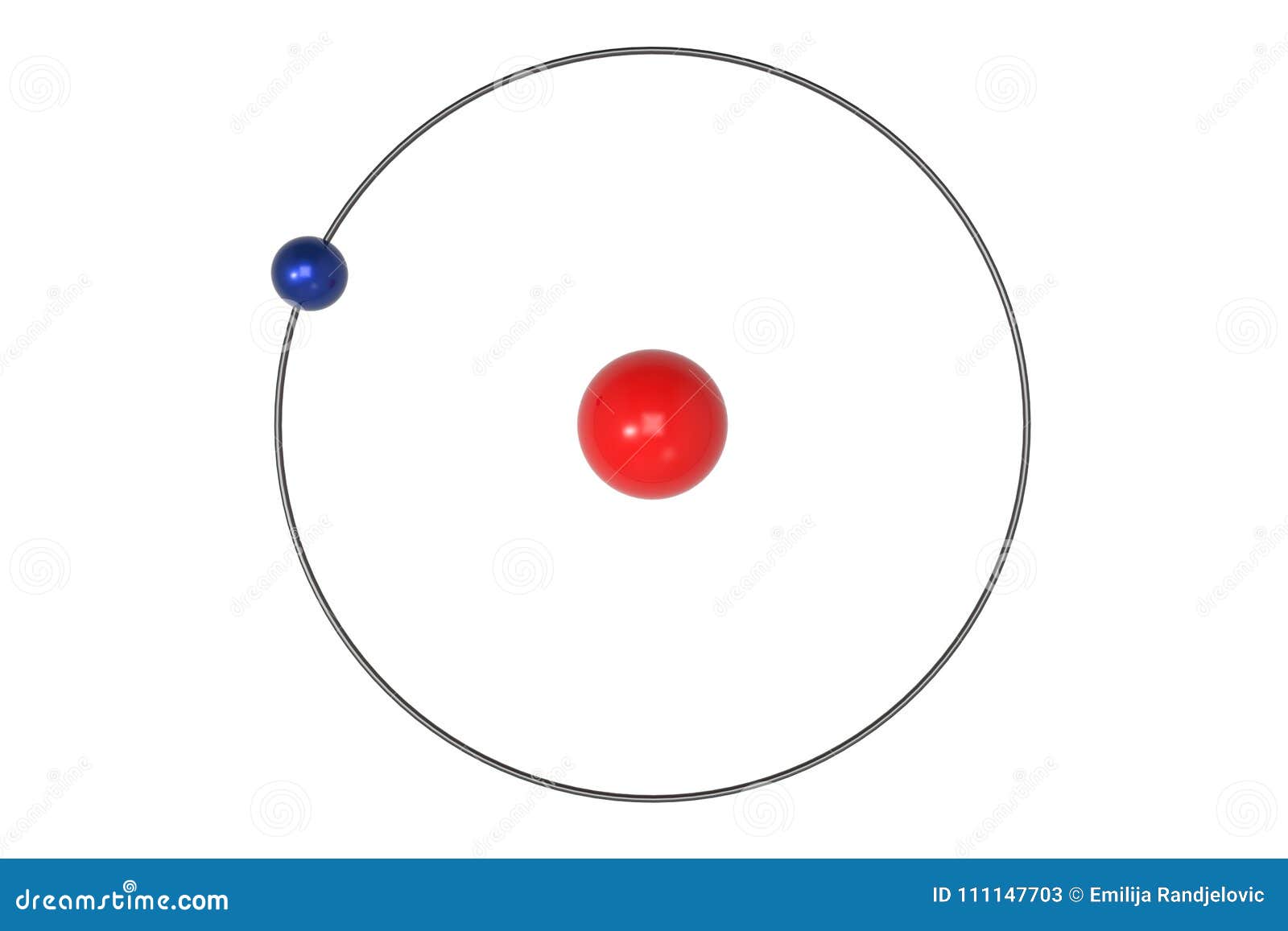
Bohr Model Hydrogen Atom My Xxx Hot Girl In 1910, arthur erich haas proposed a model of the hydrogen atom with an electron circulating on the surface of a sphere of positive charge. Bohr was able to derive the formula for the hydrogen spectrum using basic physics, the planetary model of the atom, and some very important new proposals. his first proposal is that only certain orbits are allowed: we say that the orbits of electrons in atoms are quantized. Bohr reasoned that the hydrogen atom doesn't radiate away all of its energy, because the lowest energy level (which we would now call the ground state) corresponds to exactly one wavelength fitting in the orbit, so n = 1 is the lowest it can go. It was the effort to explain this radiation that led to the first successful quantum theory of atomic structure, developed by niels bohr in 1913. he developed his theory of the hydrogenic (one electron) atom from four postulates:.
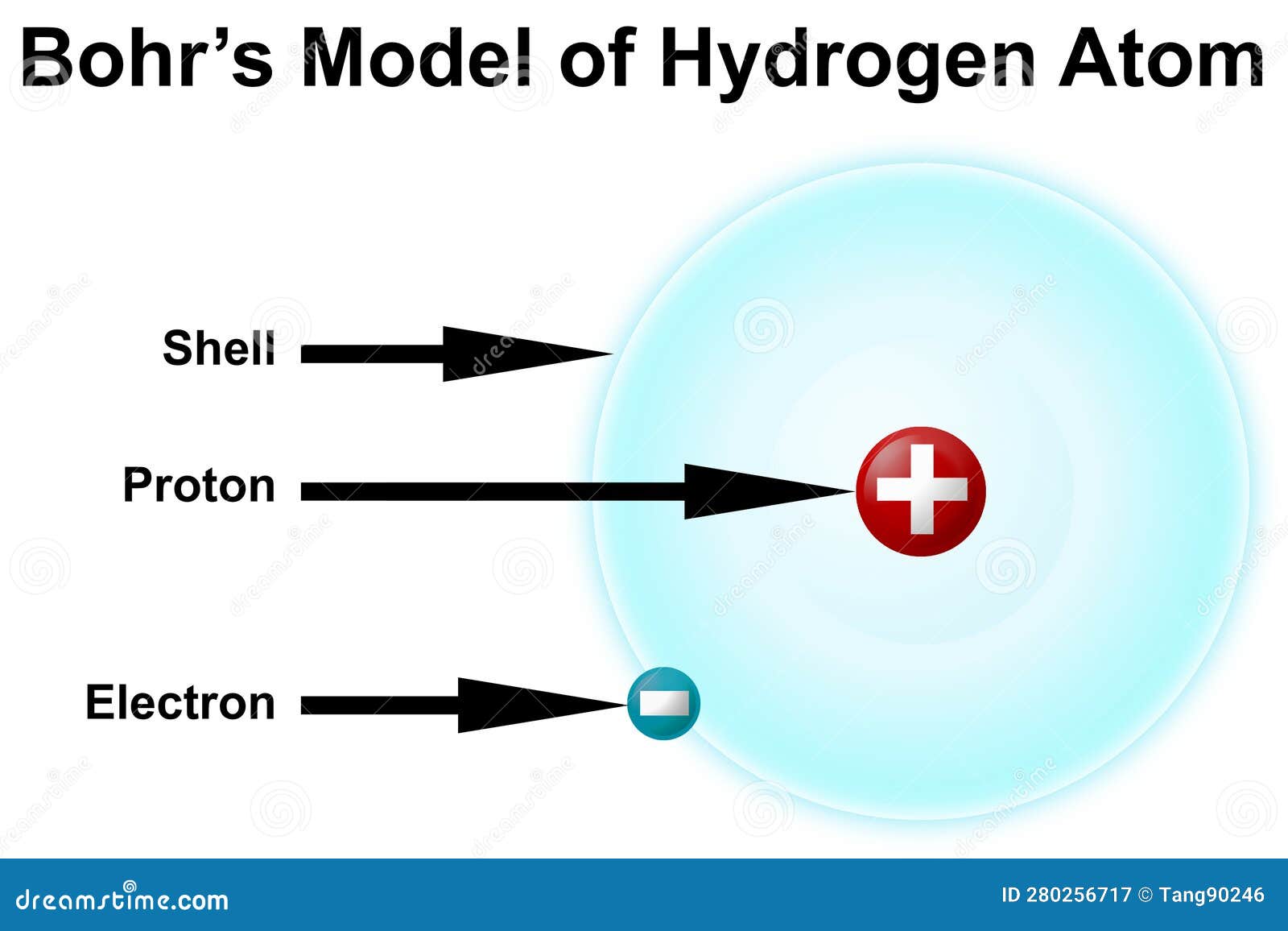
Bohr Model Of Hydrogen Atom Royalty Free Stock Photography Bohr reasoned that the hydrogen atom doesn't radiate away all of its energy, because the lowest energy level (which we would now call the ground state) corresponds to exactly one wavelength fitting in the orbit, so n = 1 is the lowest it can go. It was the effort to explain this radiation that led to the first successful quantum theory of atomic structure, developed by niels bohr in 1913. he developed his theory of the hydrogenic (one electron) atom from four postulates:. Bohr’s model of the hydrogen atom, proposed by niels bohr in 1913, was the first quantum model that correctly explained the hydrogen emission spectrum. bohr’s model combines the classical mechanics of planetary motion with the quantum concept of photons. Try out different models by shooting light at the atom. check how the prediction of the model matches the experimental results. The bohr model of the hydrogen atom (z = 1) or a hydrogen like ion (z > 1), where the negatively charged electron confined to an atomic shell encircles a small, positively charged atomic nucleus and where an electron jumps between orbits, is accompanied by an emitted or absorbed amount of electromagnetic energy (hν). [1] the orbits in which the electron may travel are shown as grey circles. Niels bohr, danish physicist, used the planetary model of the atom to explain the atomic spectrum and size of the hydrogen atom.
Comments are closed.