Biosimilars Knowledge Connect Slide Resource 1 Pdf Clinical Trial
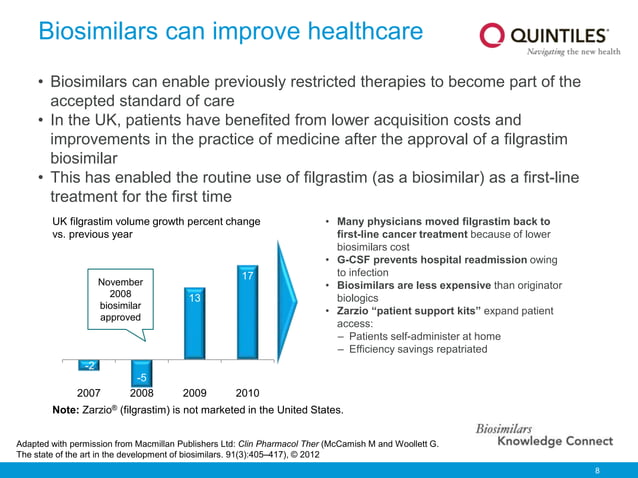
Biosimilars Knowledge Connect Slide Resource 1 Pptx This slide deck has been designed to be used as a central resource from which pertinent slides can be extracted as needed and is not intended for use in its entirety. It has been estimated that 31 different companies were developing biosimilar monoclonal antibodies (as of march 2012), compared with 18 companies as of sept 2011 – an increase of 67% in a 6 month period.1.

Biosimilars Knowledge Connect Slide Resource 1 Pptx The document provides an overview of biosimilars, which are biological medicines designed to be highly similar to their originators, impacting healthcare by potentially reducing costs. Highly regulated markets ensure safe biosimilar medicines • biosimilar quality is assured by rigorous testing requirements, which include head to head analytical non clinical clinical testing against the reference originator. The document provides an overview of biosimilars, highlighting their development, regulatory context, and the cost saving potential they offer in healthcare. The document discusses biosimilars, which are biologic medicines that are similar but not identical to an original biologic. it describes the complex multi step process used to develop and test biosimilars.

Biosimilars Knowledge Connect Slide Resource 1 Pptx The document provides an overview of biosimilars, highlighting their development, regulatory context, and the cost saving potential they offer in healthcare. The document discusses biosimilars, which are biologic medicines that are similar but not identical to an original biologic. it describes the complex multi step process used to develop and test biosimilars. Clinical trials must show comparable pharmacokinetics, pharmacodynamics and immunogenicity. while biosimilars increase access and lower costs, they are not generic copies and have unique safety profiles. This document discusses biosimilars, which are biologic medications that are similar but not identical to an original branded biologic. it notes that biosimilars are distinct from generic chemical drugs due to biological drugs' larger size and more complex structures. This document provides an overview of biosimilars including their definition, categories, development principles, and regulatory approval process. biosimilars are biological products that are highly similar to an existing approved biologic reference product. During clinical trials, patients have access to an active medicine, either biosimilar or originator. there is no placebo arm.
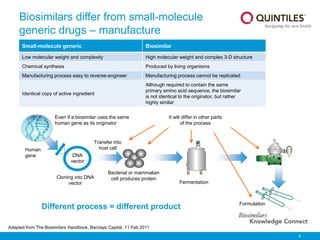
Biosimilars Knowledge Connect Slide Resource 1 Pptx Clinical trials must show comparable pharmacokinetics, pharmacodynamics and immunogenicity. while biosimilars increase access and lower costs, they are not generic copies and have unique safety profiles. This document discusses biosimilars, which are biologic medications that are similar but not identical to an original branded biologic. it notes that biosimilars are distinct from generic chemical drugs due to biological drugs' larger size and more complex structures. This document provides an overview of biosimilars including their definition, categories, development principles, and regulatory approval process. biosimilars are biological products that are highly similar to an existing approved biologic reference product. During clinical trials, patients have access to an active medicine, either biosimilar or originator. there is no placebo arm.
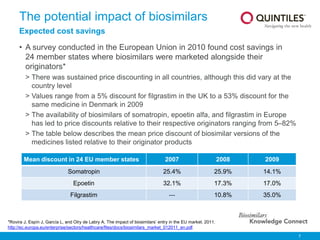
Biosimilars Knowledge Connect Slide Resource 1 Pptx This document provides an overview of biosimilars including their definition, categories, development principles, and regulatory approval process. biosimilars are biological products that are highly similar to an existing approved biologic reference product. During clinical trials, patients have access to an active medicine, either biosimilar or originator. there is no placebo arm.
Comments are closed.