Basic Chemistry Pdf Hydroxide Alkalinity

Alkalinity Pdf Alkalinity Bicarbonate The document provides an overview of acids and alkalis, detailing their properties, the ph scale, and indicators used to measure acidity and alkalinity. it explains the reactions of acids, the strength and concentration of acids and alkalis, and the formation of salts. According to arrhenius, free hydroxide ions are the most important factor in determining alkalinity in an aqueous solution.

Understanding Alkalinity Rgfinal Pdf Acid Hydroxide Weak alkalis (soluble bases) like ammonia give a ph of 10 11 but strong alkalis (soluble bases) like sodium hydroxide give a ph of 13 14. they give blue purple colour with universal indicator or litmus paper. The most important compounds in water that determine alkalinity include the carbonate (co3 2 ) and bicarbonate (hco3 ) ions. carbonate ions are able to react with and neutralize 2 hydrogen ions (h ) and the bicarbonate ions are able to neutralize h or hydroxide ions (oh ) present in water. When titrating an acid of unknown concentration with a base of known concentration, the basic solution is added to the acid solution until the equivalence point is reached. Alkalinity essentially becomes a measure of the buffering capacity of the carbonate bicarbonate ions and to some extent the hydroxide ions of water. an aqueous solution that resists changes in ph when acid or base is added is known to have sufficient buffering capacity.
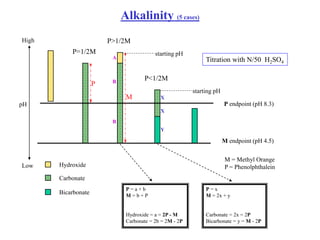
Hydroxide Alkalinity Curve This chapter will illustrate the chemistry of acid base reactions and equilibria, and provide you with tools for quantifying the concentrations of acids and bases in solutions. As ph=5, one would expect hydroxide, carbonate and bicarbonate alkalinity. the order would be: bicarbonate alkalinity > carbonate alkalinity > hydroxide alkalinity. What is alkalinity? alkalinity is the buffering capacity of a water body. it measures the ability of water bodies to neutralize acids and bases thereby maintaining a fairly stable ph. Alkalinity is the buffering capacity of a solution. it is a valuable water quality parameter used for many applications, including, but not limited to: drinking water treatment, domestic and industrial wastewater treatment, swimming pools, food and beverage, soil, agriculture, and other environmental testing.

Alkalinity Pdf Titration Chemistry What is alkalinity? alkalinity is the buffering capacity of a water body. it measures the ability of water bodies to neutralize acids and bases thereby maintaining a fairly stable ph. Alkalinity is the buffering capacity of a solution. it is a valuable water quality parameter used for many applications, including, but not limited to: drinking water treatment, domestic and industrial wastewater treatment, swimming pools, food and beverage, soil, agriculture, and other environmental testing.
Comments are closed.