Balloon Entropy

Entropy The change in entropy of the system is estimated as the change in entropy of the air in the balloon (neglecting balloon material and thin boundary layer between the balloon and room). Changes of state melting, freezing, boiling, condensing involve heat transfer , but rearranging rubber molecules from ordered to disordered has the same effect, as rob shows with a.
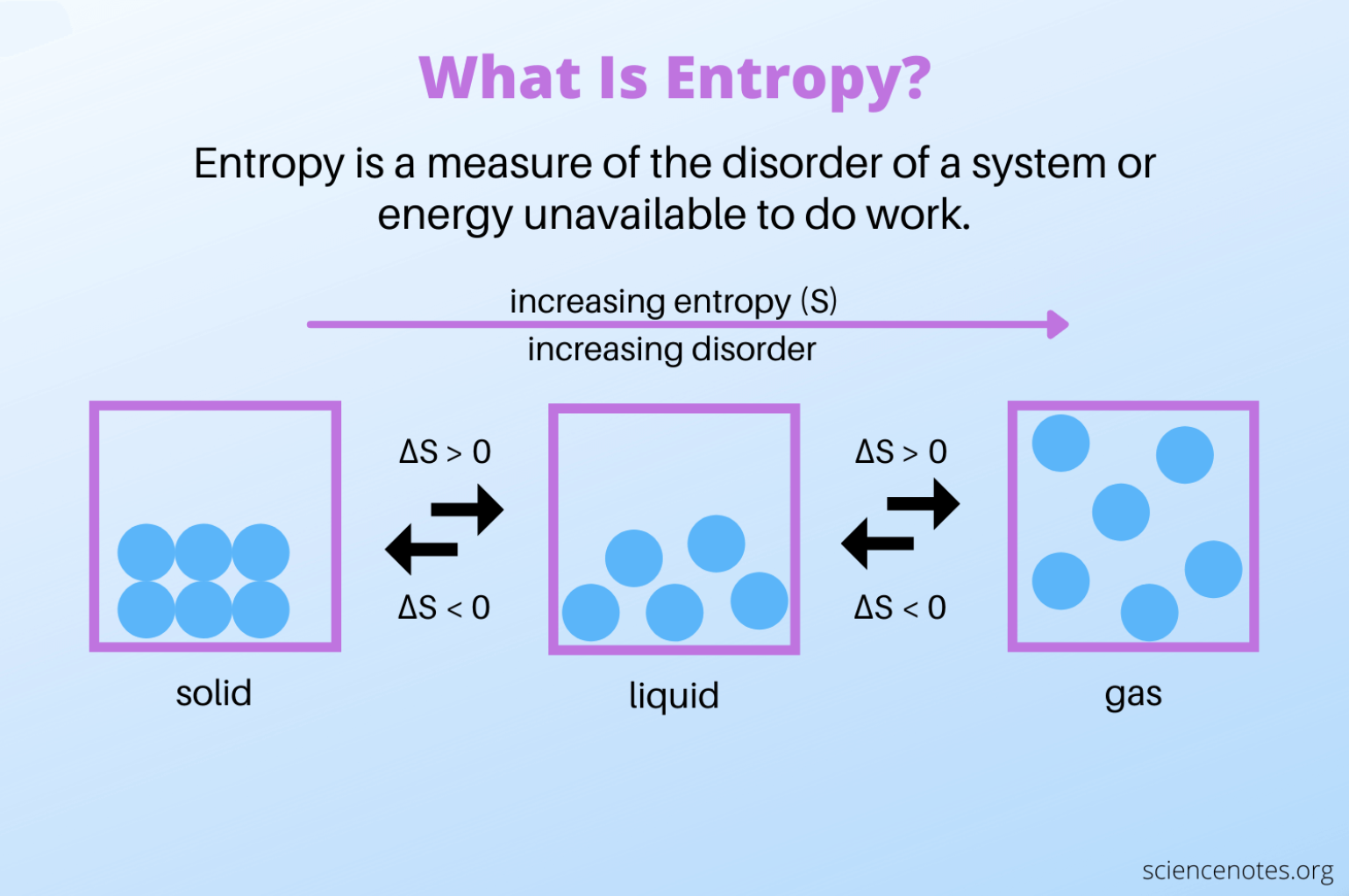
Entropy A balloon is a pure play of entropy. the entropy of the gas inside the balloon increases when the balloon expands, but the entropy of the polymer network of the membrane increases when the balloon contracts. Balloon model directly maps ‘pressure’ to ‘entropy’ for instant intuition. show the entropy gradient ∇ₓs (analog of −∂f ∂v) as a concrete formula for physical intuition. The behaviour of a blown up balloon can be explained by entropy, which is often referred to as disorder. the second law of thermodynamics states that the entropy in an isolated system will increase, and spontaneous change is associated with an increase in entropy. The first law tells us that all the work we’ve done stretching the balloon has gone directly into internal stored energy in the balloon. the second law tells us that if the entropy decreased, the temperature has to increase!.

Upgrade Entropy Feed The Beast Wiki The behaviour of a blown up balloon can be explained by entropy, which is often referred to as disorder. the second law of thermodynamics states that the entropy in an isolated system will increase, and spontaneous change is associated with an increase in entropy. The first law tells us that all the work we’ve done stretching the balloon has gone directly into internal stored energy in the balloon. the second law tells us that if the entropy decreased, the temperature has to increase!. The discussion revolves around calculating the total entropy of a helium balloon when transitioning from one temperature to another. the context involves thermodynamics and the behavior of ideal gases. Entropy is a measure of disorder, dispersal, and the number of microstates a system can occupy. learn how entropy relates to particle arrangements, phase changes, and probability in chemical systems. This expression gives entropy change in terms of temperature and volume. we can develop an alternative form in terms of pressure and volume, which allows us to examine an assumption we have used. With just a little bit of baking soda and vinegar, you can create a chemical reaction that will actually do all the blowing you need. this is also a good way to measure the molarity (moles per liter) of a substance.

Entropy An Immersive Visual Journey Of Light Art And Nature S Wonders The discussion revolves around calculating the total entropy of a helium balloon when transitioning from one temperature to another. the context involves thermodynamics and the behavior of ideal gases. Entropy is a measure of disorder, dispersal, and the number of microstates a system can occupy. learn how entropy relates to particle arrangements, phase changes, and probability in chemical systems. This expression gives entropy change in terms of temperature and volume. we can develop an alternative form in terms of pressure and volume, which allows us to examine an assumption we have used. With just a little bit of baking soda and vinegar, you can create a chemical reaction that will actually do all the blowing you need. this is also a good way to measure the molarity (moles per liter) of a substance.
Comments are closed.