Atoms And Ions Unveiling Their Differences

Differentiating Atoms Molecules And Ions Stem Gc11am Ic E 20 Pdf Atoms are the basic units of matter, stable and neutral, composed of protons, neutrons, and electrons. ions, however, are atoms charged through the loss or gain of electrons. this fundamental difference is critical for understanding chemical reactions and properties. The atoms in chemical compounds are held together by attractive electrostatic interactions known as chemical bonds. ionic compounds contain positively and negatively charged ions in a ratio that results in an overall charge of zero.

Lesson 3 Atoms Molecules And Ions Pdf Atoms and ions are fundamental particles that dictate how matter interacts and behaves. this article will clarify the differences between these two concepts in chemistry. While atoms are the basic building blocks of all matter, ions are atoms that have gained or lost electrons, resulting in a net positive or negative charge. in this article, we will explore the attributes of atoms and ions, highlighting their similarities and differences. Ions are atoms or molecules that have gained or lost one or more electrons, acquiring an electric charge. when an atom loses electrons, it becomes a cation, which is a positively charged ion. when an atom gains electrons, it becomes an anion, which is a negatively charged ion. Learn the difference between atom and ion with clear examples and a comparison table. a quick chemistry guide for students and curious learners.

Atoms Vs Ions Play Online On Flash Museum рџ пёџ Ions are atoms or molecules that have gained or lost one or more electrons, acquiring an electric charge. when an atom loses electrons, it becomes a cation, which is a positively charged ion. when an atom gains electrons, it becomes an anion, which is a negatively charged ion. Learn the difference between atom and ion with clear examples and a comparison table. a quick chemistry guide for students and curious learners. Under this broader definition, an atom can be an ion, but not all ions are atoms. the difference between an atom and an ion has to do with net electrical charge. examples of atoms include hydrogen (h), helium (he), iron (fe), and any other element on the periodic table. When an atom's outermost orbital gains or loses electrons (also known as valence electrons), the atom forms an ion. an ion with more protons than electrons carries a net positive charge and is called a cation. Each element is composed of extremely small particles called atoms. all atoms of a given element are identical to one another in mass and other properties, but the atoms of one element are different from the atoms of all other elements. In its stable form, an atom has an equal number of protons and electrons, resulting in a neutral charge. on the other hand, an ion is formed when an atom or molecule either loses or gains electrons, leading to an imbalance between the number of protons and electrons and thus a net electric charge.
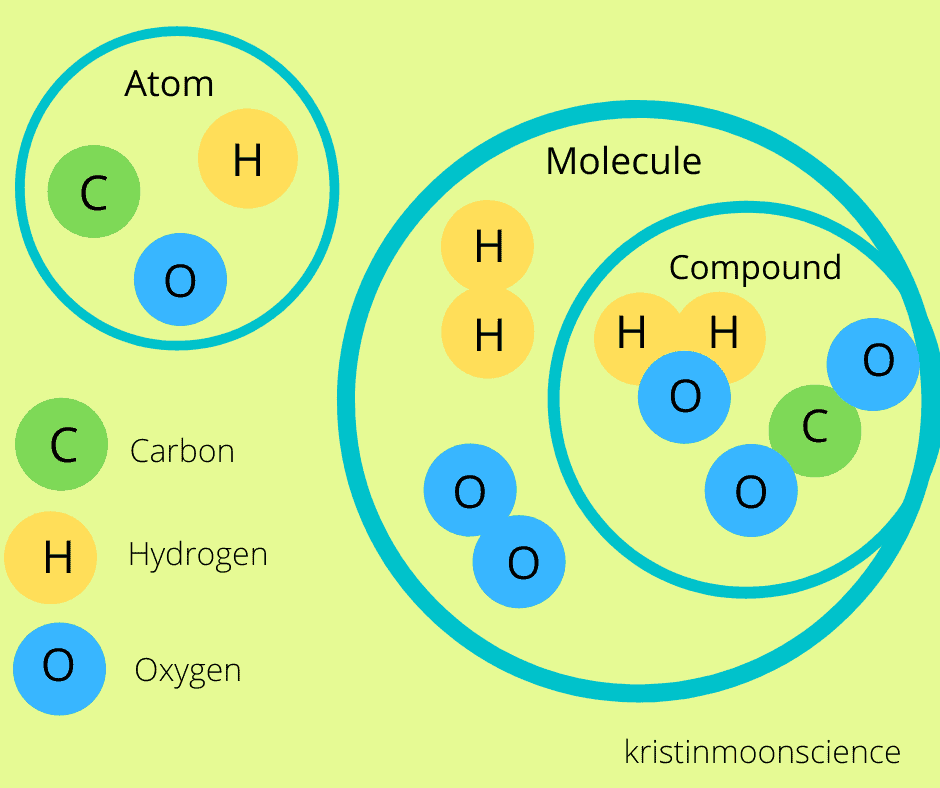
Atoms And Ions Difference Between Atoms And Ions Explanation Under this broader definition, an atom can be an ion, but not all ions are atoms. the difference between an atom and an ion has to do with net electrical charge. examples of atoms include hydrogen (h), helium (he), iron (fe), and any other element on the periodic table. When an atom's outermost orbital gains or loses electrons (also known as valence electrons), the atom forms an ion. an ion with more protons than electrons carries a net positive charge and is called a cation. Each element is composed of extremely small particles called atoms. all atoms of a given element are identical to one another in mass and other properties, but the atoms of one element are different from the atoms of all other elements. In its stable form, an atom has an equal number of protons and electrons, resulting in a neutral charge. on the other hand, an ion is formed when an atom or molecule either loses or gains electrons, leading to an imbalance between the number of protons and electrons and thus a net electric charge.
Comments are closed.