Atomic Structure Notes Pdf Atomic Physics Radiation
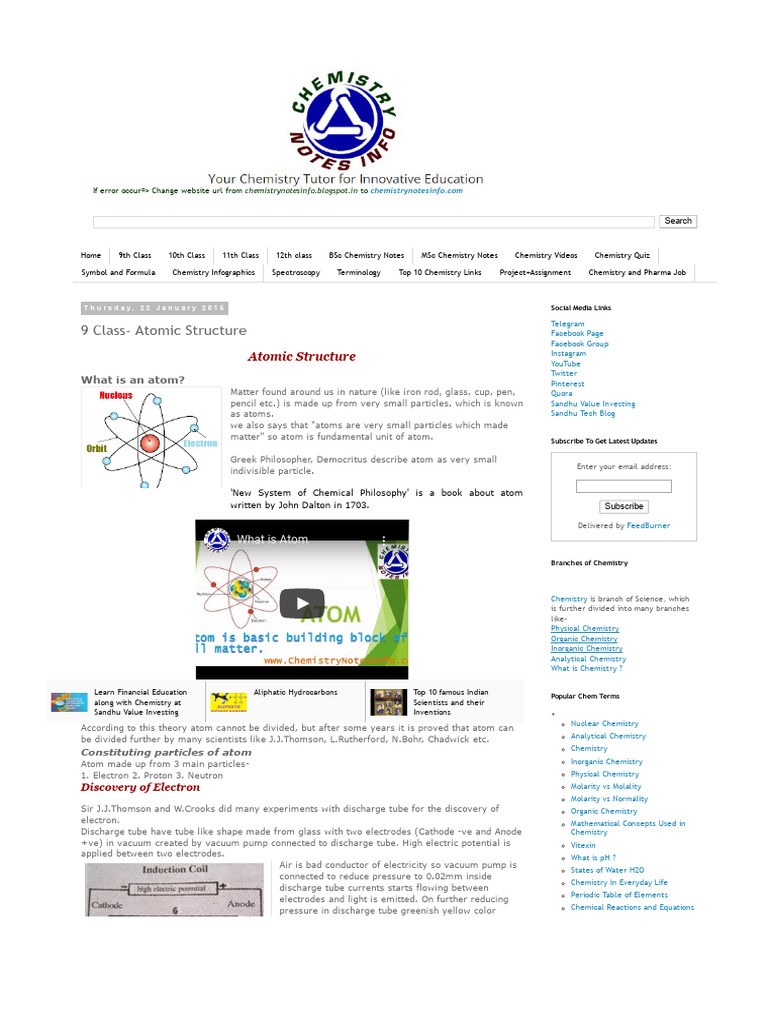
9 Class Atomic Structure Notes Pdf Download Free Pdf Atoms Atomic physics underlies the study of astrophysics and solid state physics. it has led to important applications in medicine, communications, lasers etc, as well as still providing a testing ground for quantum theory and its derivatives, quantum electrodynamics. It explains concepts like isotopes, radioisotopes, types of radiation (alpha, beta, gamma), and the process of radioactive decay, including half life and ionization. additionally, it discusses the implications of radiation on health and safety precautions necessary when handling radioactive materials.

Atomic Structure Notes Pdf Nuclear physics welcome to the fascinating world of atomic structure! today, we’re going to explore the nucleus, the tiny centre of the atom, and learn about radioactivity. An element may be composed of atoms that all have the same number of protons, but have different numbers of neutrons, i.e. have different atomic mass numbers a. atoms of identical atomic number z but differing atomic mass numbers a are called isotopes of a given element. Atoms of the same element have the same atomic number and number of protons. an atom has no charge overall so the number of electrons must equal to the number of protons to cancel the charges. Like the electrons in an excited atom, the nucleons in an excited nucleus will transition towards their lowest energy configuration and in doing so emit a discrete bundle of electromagnetic radiation called a gamma ray (g ray).

Atomic Structure Notes Pdf Atoms Proton Atoms of the same element have the same atomic number and number of protons. an atom has no charge overall so the number of electrons must equal to the number of protons to cancel the charges. Like the electrons in an excited atom, the nucleons in an excited nucleus will transition towards their lowest energy configuration and in doing so emit a discrete bundle of electromagnetic radiation called a gamma ray (g ray). When a californium atom reacts with an unknown element x, the following reaction occurs. it is important to be familiar the properties of each type of radiation and their symbols. This series of supplementary notes (sni to snvi) presents all the topics in the nuclear and radiation (n&r) section of phy138. the material in the appendices is provided for interest or and to aid your understanding; it is not, however, examinable. Before you learn about the internal structure of an atom and the experiments that led to their discovery, it is important to outline key concepts in electric charge and about the discovery of ions. Niels bohr in 1913 combined rutherford’s concept of the nuclear atom with planck’s idea of the quantized nature of the radiation process and developed an atomic model that successfully deals with one electron structures, such as the hydrogen atom, singly ionized helium, etc.

Atomic Structure Notes Kp Lh Pdf Atoms Atomic Nucleus When a californium atom reacts with an unknown element x, the following reaction occurs. it is important to be familiar the properties of each type of radiation and their symbols. This series of supplementary notes (sni to snvi) presents all the topics in the nuclear and radiation (n&r) section of phy138. the material in the appendices is provided for interest or and to aid your understanding; it is not, however, examinable. Before you learn about the internal structure of an atom and the experiments that led to their discovery, it is important to outline key concepts in electric charge and about the discovery of ions. Niels bohr in 1913 combined rutherford’s concept of the nuclear atom with planck’s idea of the quantized nature of the radiation process and developed an atomic model that successfully deals with one electron structures, such as the hydrogen atom, singly ionized helium, etc.
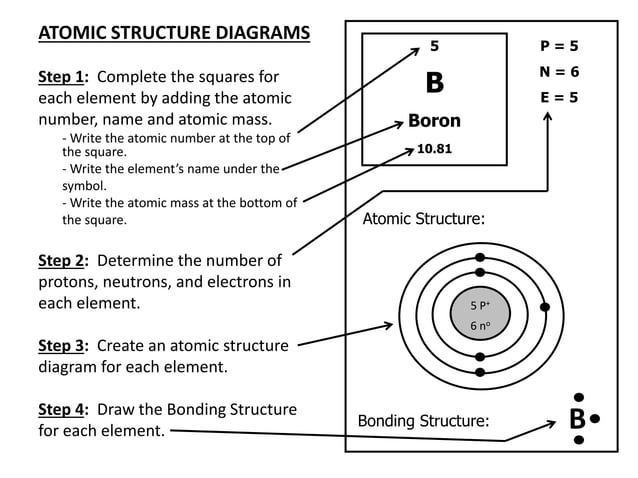
Atomic Structure Notes Pdf Chemistry Science Before you learn about the internal structure of an atom and the experiments that led to their discovery, it is important to outline key concepts in electric charge and about the discovery of ions. Niels bohr in 1913 combined rutherford’s concept of the nuclear atom with planck’s idea of the quantized nature of the radiation process and developed an atomic model that successfully deals with one electron structures, such as the hydrogen atom, singly ionized helium, etc.

Pdf Atomic Structure Handwritten Notes 2 Compress Pdf
Comments are closed.