Atomic Structure Bohr S Model Presentation
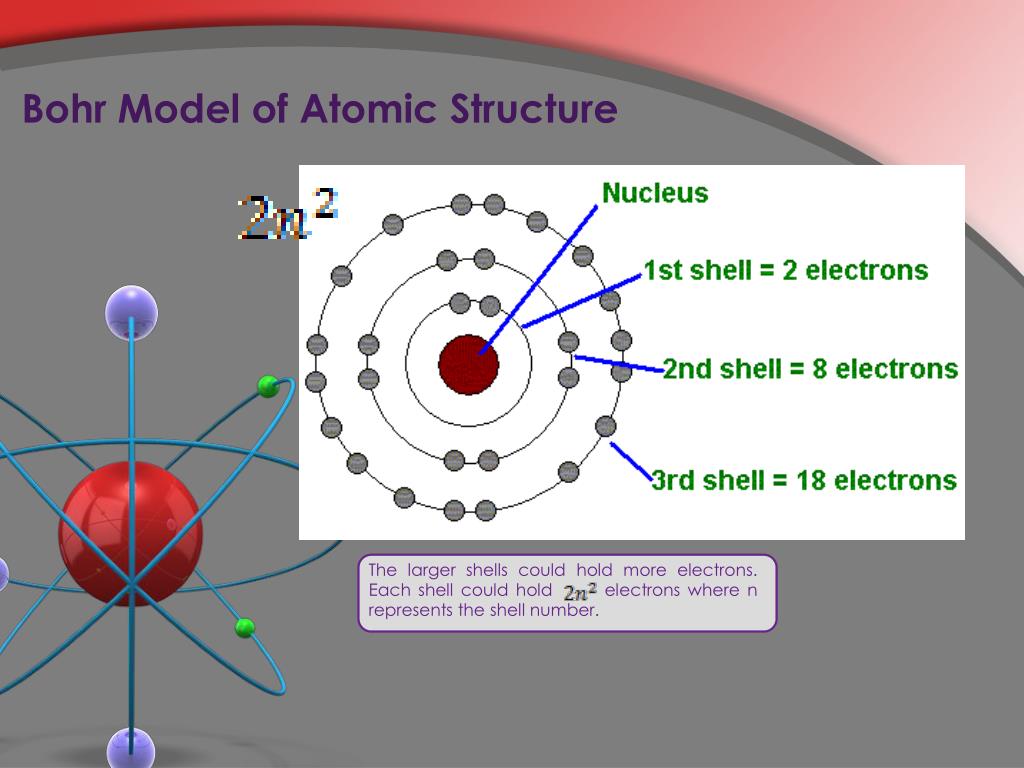
Bohr Model Atomic Structure The document outlines the development of atomic models, starting with democritus's early idea of atoms, followed by dalton's atomic theory and thomson's 'plum pudding' model which introduced electrons. Bohr's atomic model explained the document discusses the development of atomic structure models from thomson to bohr. j.j. thomson's "plum pudding" model was replaced by rutherford's nuclear model based on his gold foil experiment.
:max_bytes(150000):strip_icc()/atomic-structure-conceptual-artwork-99312661-58af58c75f9b5860467ff472.jpg)
Bohr Model Atomic Structure Electrons examples: bohr model 1913 – bohr explains movement of electrons around the nucleus of the atom. bohr’s model: electrons orbit around the nucleus. This overview traces the transformation of atomic theory, beginning with niels bohr's model based on hydrogen. bohr's planetary model proposed quantized energy levels for electrons, akin to rings of an onion. while successful for hydrogen, it paved the way for quantum mechanical advancements . In the present first part of the paper the mechanism of the binding of electrons by a positive nucleus is discussed in relation to planck’s theory. it will be shown that it is possible from the point of view taken to account in a simple way for the law of the line spectrum of hydrogen. niels bohr, 1913. Simple models of the atom now included the nucleus with the protons and neutrons and the energy levels with the electrons. bohr models are drawn similar to the images shown here.

Bohr Model Of Atomic Structure Assefo In the present first part of the paper the mechanism of the binding of electrons by a positive nucleus is discussed in relation to planck’s theory. it will be shown that it is possible from the point of view taken to account in a simple way for the law of the line spectrum of hydrogen. niels bohr, 1913. Simple models of the atom now included the nucleus with the protons and neutrons and the energy levels with the electrons. bohr models are drawn similar to the images shown here. According to the bohr model, which of the ions listed in the table below would have the smallest distance between an electron in the lowest energy level and the nucleus?. In 1913, niels bohr proposed a theoretical model for the hydrogen atom that explained its emission spectrum. – his model required only one assumption: the electron moves around the nucleus in circular orbits that can have only certain allowed radii. 7 how many electrons fit in the second orbit? 9p 10n bohr's model of the atom e.g. fluorine: #p = 9 #e = 9 #n = 10 how many electrons are left?. In a classic paper published in 1913, the young niels bohr, then working with rutherford in manchester, england, proceeded to show how rydberg’s formula could be explained in terms of a very simple model of the hydrogen atom.

Atomic Structure Bohr S Model Of Atom Pdf According to the bohr model, which of the ions listed in the table below would have the smallest distance between an electron in the lowest energy level and the nucleus?. In 1913, niels bohr proposed a theoretical model for the hydrogen atom that explained its emission spectrum. – his model required only one assumption: the electron moves around the nucleus in circular orbits that can have only certain allowed radii. 7 how many electrons fit in the second orbit? 9p 10n bohr's model of the atom e.g. fluorine: #p = 9 #e = 9 #n = 10 how many electrons are left?. In a classic paper published in 1913, the young niels bohr, then working with rutherford in manchester, england, proceeded to show how rydberg’s formula could be explained in terms of a very simple model of the hydrogen atom.
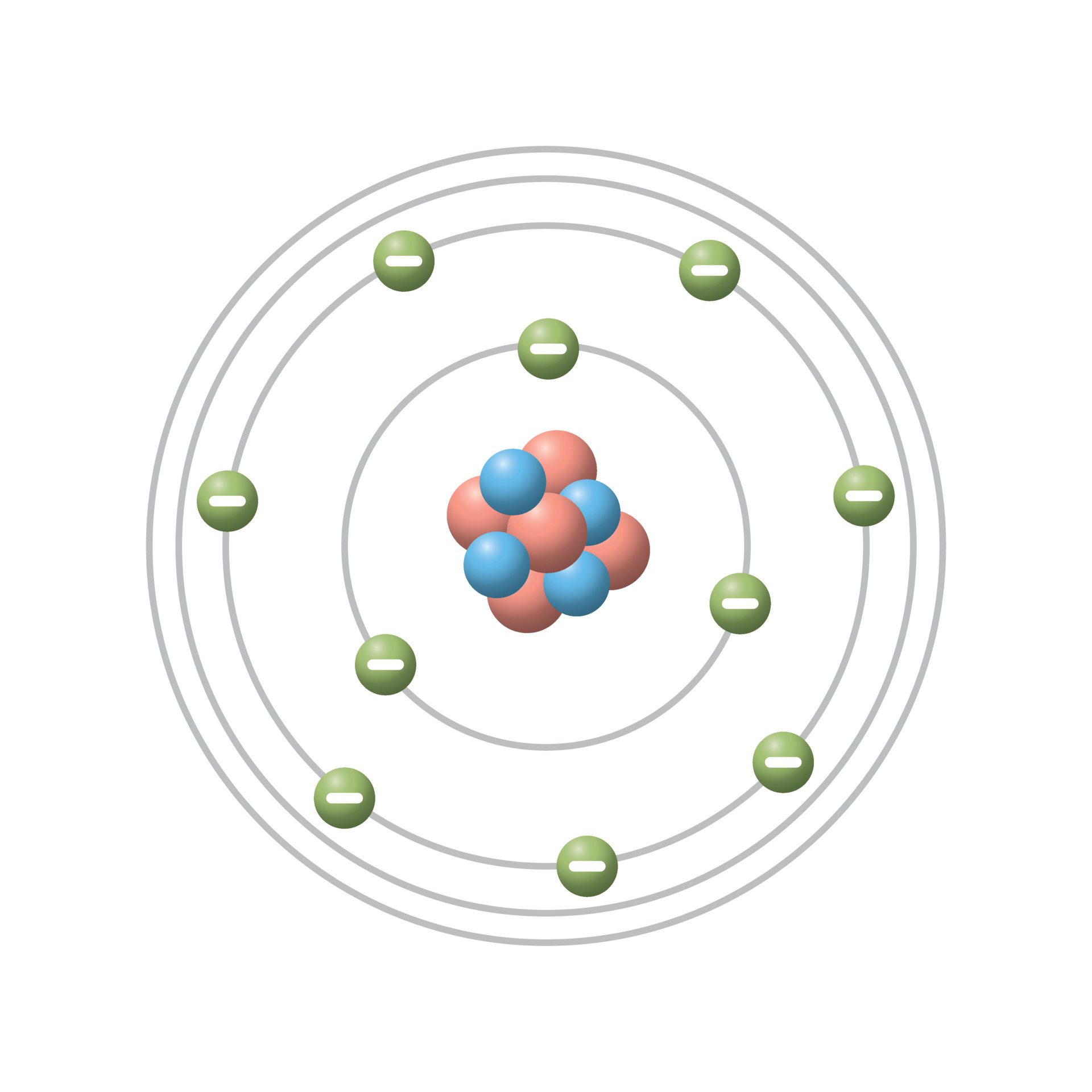
Vector Bohr Model Description Of The Structure Of Atoms 52 Off 7 how many electrons fit in the second orbit? 9p 10n bohr's model of the atom e.g. fluorine: #p = 9 #e = 9 #n = 10 how many electrons are left?. In a classic paper published in 1913, the young niels bohr, then working with rutherford in manchester, england, proceeded to show how rydberg’s formula could be explained in terms of a very simple model of the hydrogen atom.
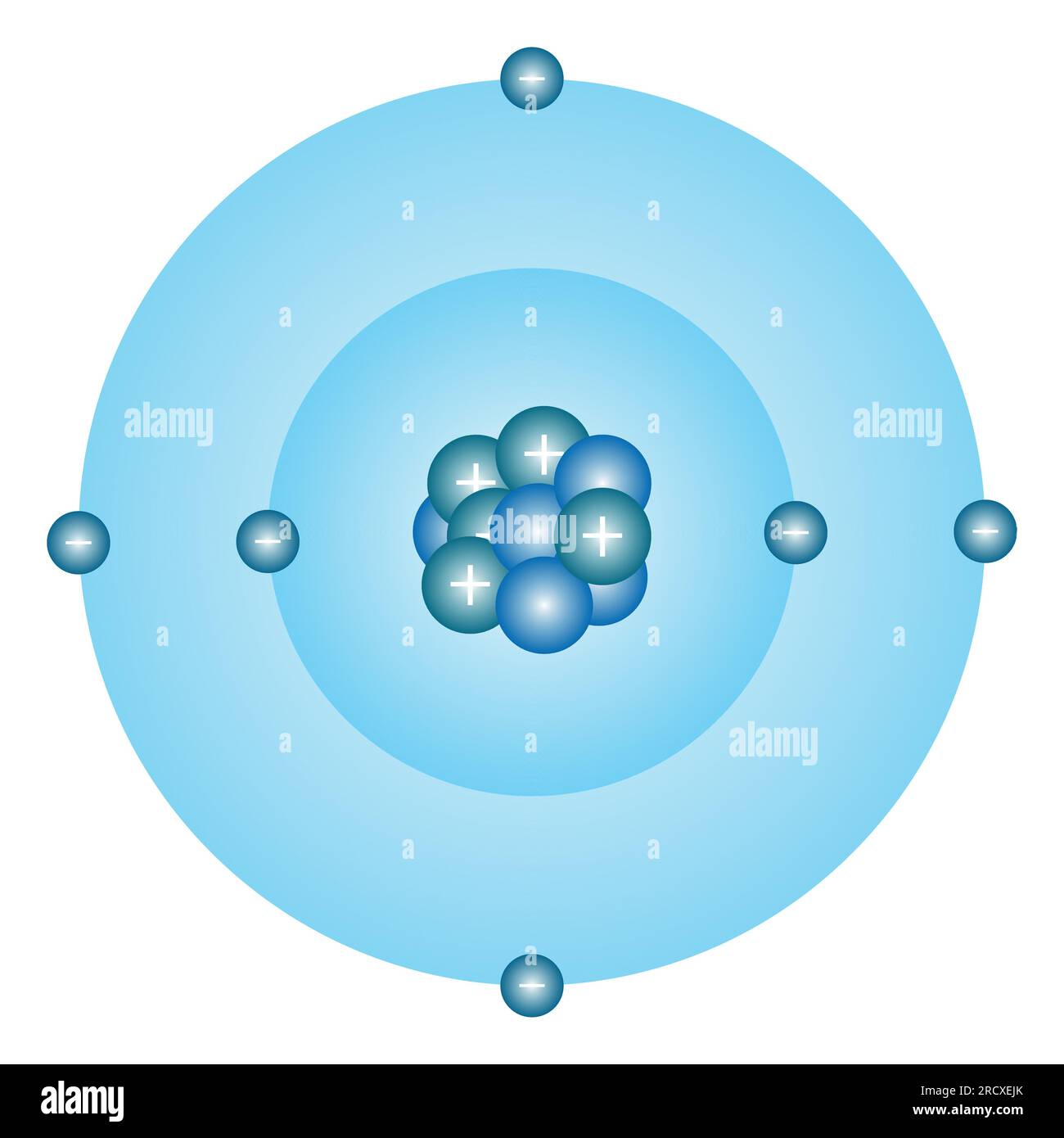
Bohr Atomic Model Of Atom Proton Neutron Electron And Electron
Comments are closed.