Atomic Structure Bohr S Atomic Model Most Important For Iit Jee And

Bohr S Atomic Model Jee 11 Pdf Electron Atoms This document provides comprehensive notes on the structure of the atom, covering subatomic particles, atomic models, and key theories such as bohr's model and the quantum mechanical model. Boost your jee main chemistry prep with solved previous year questions on atomic structure. practice mcqs on bohr’s model, quantum numbers, and de broglie equation with expert solutions and detailed explanations.

Iit Jee Atomic Structure Ans Pdf Electron Atoms Ans. bohr's atomic model is a theoretical framework proposed by physicist niels bohr to describe the structure of atoms. it introduced the concept of quantized energy levels for electrons orbiting the nucleus and explained atomic spectra, particularly the discrete line spectrum of hydrogen. Discover bohr’s atomic model with easy postulates, energy levels, diagrams, and real life examples. master concepts for neet, jee, and board exams. Physical chemistry by o p tandon is a must have for all those who are preparing for iit jee or other engineering entrance examinations.he book covers the entire physical chemistry syllabus including atomic structure, radioactivity and nuclear transformation and stoichiometry. In 1913, niels bohr proposed a revolutionary model of the atom that successfully explained the hydrogen spectrum. while later superseded by quantum mechanics, bohr’s model remains essential for jee and provides intuitive understanding of atomic structure.
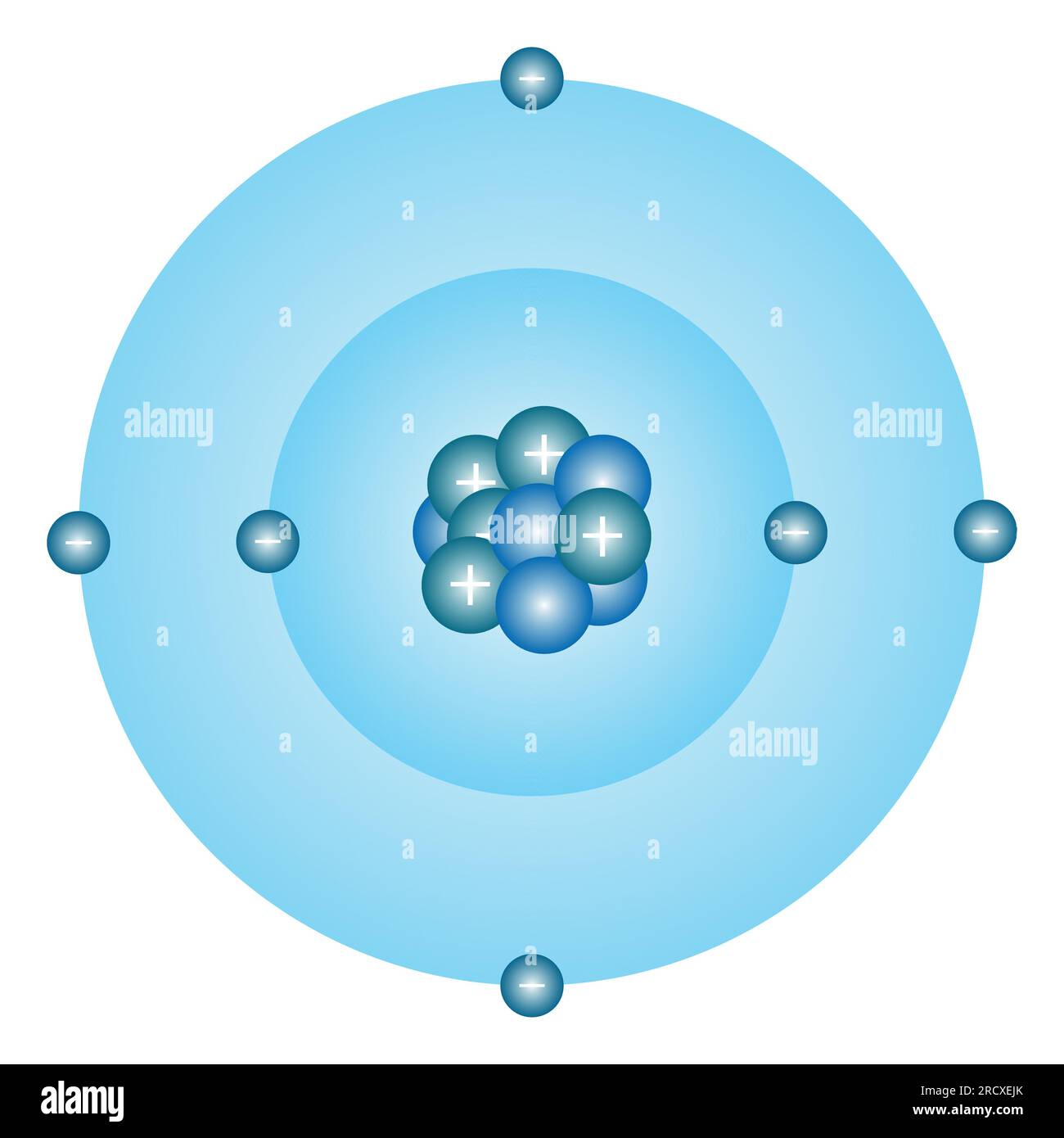
Jee Atomic Structure L Bohr S Model Of Atom Unacademy Jee Jee My Xxx Physical chemistry by o p tandon is a must have for all those who are preparing for iit jee or other engineering entrance examinations.he book covers the entire physical chemistry syllabus including atomic structure, radioactivity and nuclear transformation and stoichiometry. In 1913, niels bohr proposed a revolutionary model of the atom that successfully explained the hydrogen spectrum. while later superseded by quantum mechanics, bohr’s model remains essential for jee and provides intuitive understanding of atomic structure. Neil bohr explained the line spectrum while developing the model of an atom. we discuss the various postulates of the bohr atomic model. the bohr model shows that the electrons in atoms are in orbits of differing energy around the nucleus. By the early 20th century, rutherford's model gave us a picture of the atom: a tiny, dense, positive nucleus with electrons orbiting it. but classical physics predicted these orbiting electrons should continuously radiate energy, spiral inwards, and collapse into the nucleus. In this introduction to atomic structure, learn the development, impact and limitations of bohr’s atomic theory. In this article, we will cover the concept of bohr's model and its postulates. this concept falls under the broader category of atomic structure, which is a crucial chapter in class 11 chemistry.

Iit Jee Atomic Structure Apex Classes Neil bohr explained the line spectrum while developing the model of an atom. we discuss the various postulates of the bohr atomic model. the bohr model shows that the electrons in atoms are in orbits of differing energy around the nucleus. By the early 20th century, rutherford's model gave us a picture of the atom: a tiny, dense, positive nucleus with electrons orbiting it. but classical physics predicted these orbiting electrons should continuously radiate energy, spiral inwards, and collapse into the nucleus. In this introduction to atomic structure, learn the development, impact and limitations of bohr’s atomic theory. In this article, we will cover the concept of bohr's model and its postulates. this concept falls under the broader category of atomic structure, which is a crucial chapter in class 11 chemistry.

Atomic Structure Preparation Tips For Iit Jee Askiitians In this introduction to atomic structure, learn the development, impact and limitations of bohr’s atomic theory. In this article, we will cover the concept of bohr's model and its postulates. this concept falls under the broader category of atomic structure, which is a crucial chapter in class 11 chemistry.
Comments are closed.