Antibody Drug Conjugates Characterization

Antibody Drug Conjugates Characterization Waters Videos In this review, analytical methods for physicochemical characterization of adcs will be reported and analysed in order to highlight as the chromatographic procedures allow obtaining a complete adcs evaluation and characterization. Abstract antibody drug conjugates (adcs) comprise a monoclonal antibody covalently bound to a cytotoxic payload by a linker. adcs minimize off target effects on healthy tissues, leveraging the specificity of monoclonal antibodies to deliver cytotoxic drugs to the intended tumor site.
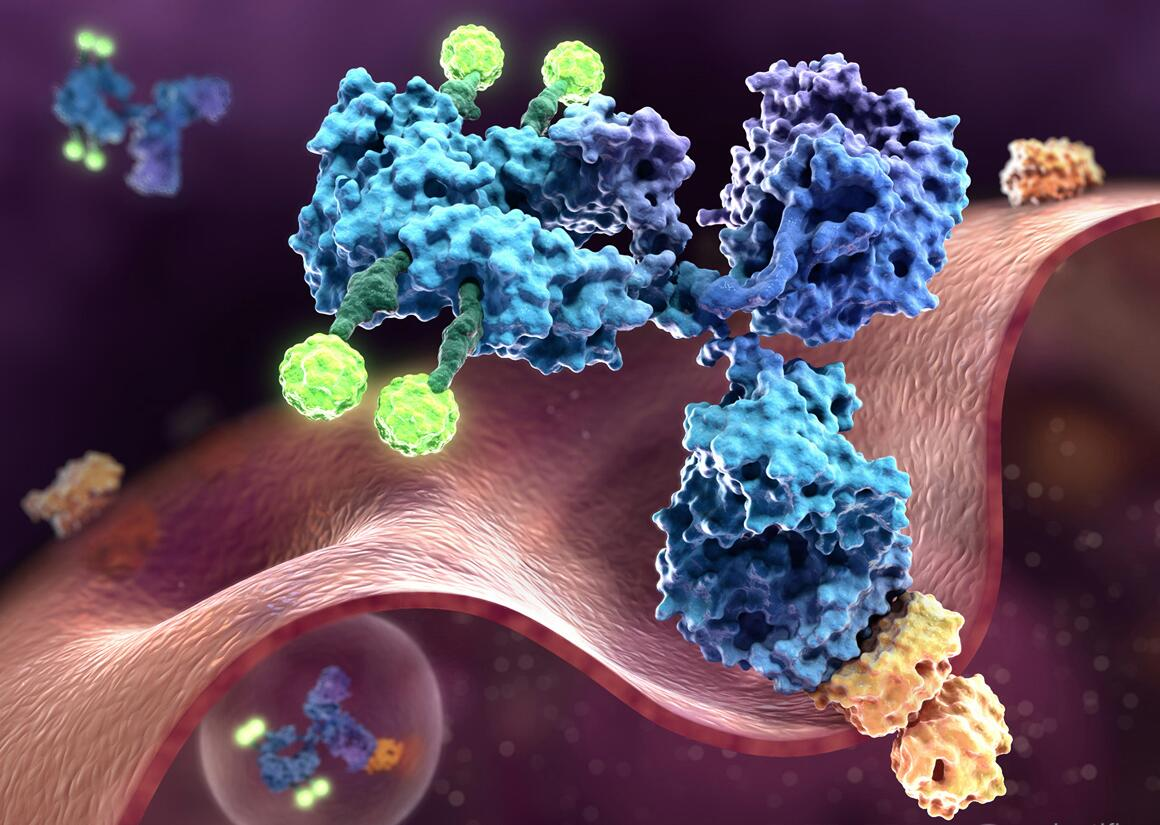
Antibody Drug Conjugates Characterization Service Labinsights Antibody–drug conjugates utilize the high target affinity of antibodies to bind and release their cytotoxic payload at the tumor site. however, in order to create an effective treatment, biophysical characterization of antibody–drug conjugates is essential. Antibody drug conjugates (adcs) are transforming the treatment landscape for cancer and other complex diseases by combining the specificity of monoclonal antibodies with the potency of cytotoxic drugs. Scaling antibody drug conjugate characterization with lab automation shweta shukradas from agilent technologies discusses how automation and instrumentation advances enable faster and more reproducible targeted biologic development. Before they may be more widely used, several obstacles need to be cleared. this review discusses mabs, adcs, acnps, various conjugation strategies, characterization techniques, and current status and bottlenecks for both concepts.

Improved Characterization Of Antibody Drug Conjugates Technology Networks Scaling antibody drug conjugate characterization with lab automation shweta shukradas from agilent technologies discusses how automation and instrumentation advances enable faster and more reproducible targeted biologic development. Before they may be more widely used, several obstacles need to be cleared. this review discusses mabs, adcs, acnps, various conjugation strategies, characterization techniques, and current status and bottlenecks for both concepts. Figure 1. a schematic representation of an antibody–drug conjugate. the mab portion of an adc consists of two light chains (green) and two heavy chains (blue), covalently linked by disulfide bonds. the antigen binding site is located in the variable domains (vl and vh), which each bear three loops that together comprise the complementarity determining region (cdr). cytotoxic payloads (purple. As a result, appropriate characterization of candidate molecules using different analytical techniques is often required to select optimal adcs and to obtain a complete characterization profile during development. Mentioning: 2 new analytical challenges in characterization of antibody drug conjugates ricci, erika maria, dainese, enrico, de laurenzi, vincenzo, lovato, giulio. This review describes the development, characterization, and regulatory consideration of adcs, and it summarizes the approved products in the market and in clinical trials.
Illustration Of Antibody Drug Conjugates Antibody Drug Conjugates Can Figure 1. a schematic representation of an antibody–drug conjugate. the mab portion of an adc consists of two light chains (green) and two heavy chains (blue), covalently linked by disulfide bonds. the antigen binding site is located in the variable domains (vl and vh), which each bear three loops that together comprise the complementarity determining region (cdr). cytotoxic payloads (purple. As a result, appropriate characterization of candidate molecules using different analytical techniques is often required to select optimal adcs and to obtain a complete characterization profile during development. Mentioning: 2 new analytical challenges in characterization of antibody drug conjugates ricci, erika maria, dainese, enrico, de laurenzi, vincenzo, lovato, giulio. This review describes the development, characterization, and regulatory consideration of adcs, and it summarizes the approved products in the market and in clinical trials.
Comments are closed.