Antibody Drug Conjugates Bioanalysis Zone
Antibody Drug Conjugates Bioanalysis Zone Welcome to the bioanalysis zone spotlight on antibody drug conjugates (adcs)! adcs are dynamic, heterogeneous mixtures typically consisting of a cytotoxic drug covalently bound to a monoclonal antibody via a synthetic linker. Antibody–drug conjugates (adcs) are a new class of biotherapeutics, consisting of a cytotoxic payload covalently bound to an antibody by a linker. ligand binding assay (lba) and liquid chromatography mass spectrometry (lc ms) are the favored techniques for the analysis of adcs in biomatrices.
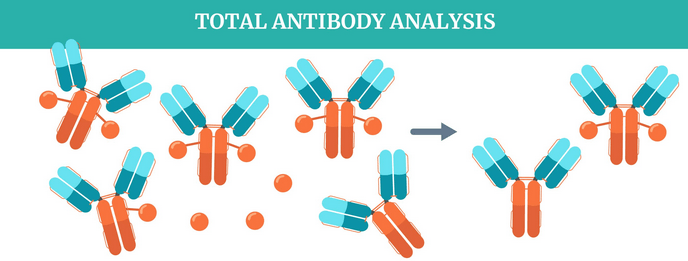
In The Zone Antibody Drug Conjugates Bioanalysis Zone The components of adcs, such as total antibody, conjugated antibody, conjugated payload, and free payload, need to be determined. in addition, monitoring changes in dar in vivo offers critical information for evaluating adc pharmacokinetics. Here, we provide the different bioanalytical strategies to determine the pharmacokinetics and immunogenicity of adc molecules. Since 2001 qps’ bioanalytical teams have contributed to antibody drug conjugate (adc) drug development, supporting the filing of one of the first drug targeting programs and continuing to develop customized strategies for novel conjugate molecules. Antibody drug conjugates (adcs), primarily used in anti cancer therapy, offer a promising treatment alternative due to their ability to deliver a therapeutic molecule while simultaneously.
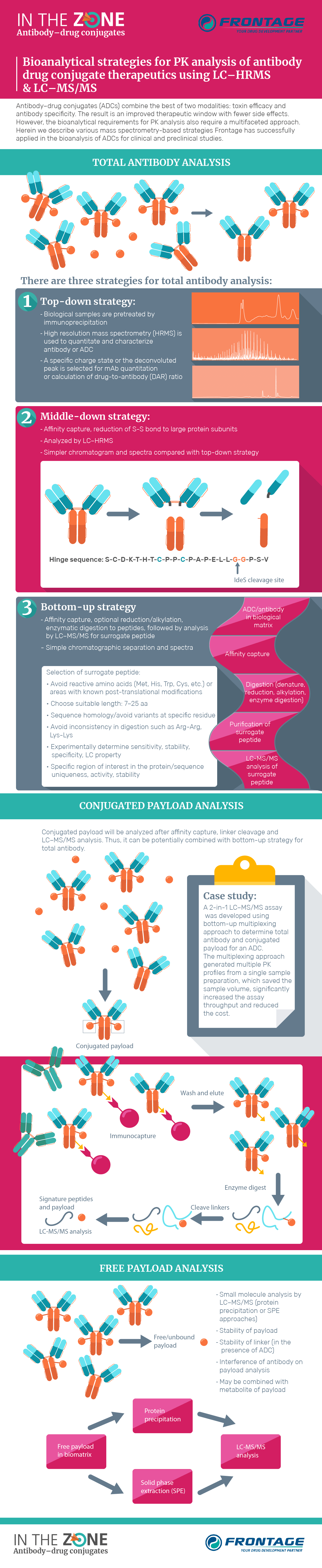
In The Zone Antibody Drug Conjugates Bioanalysis Zone Since 2001 qps’ bioanalytical teams have contributed to antibody drug conjugate (adc) drug development, supporting the filing of one of the first drug targeting programs and continuing to develop customized strategies for novel conjugate molecules. Antibody drug conjugates (adcs), primarily used in anti cancer therapy, offer a promising treatment alternative due to their ability to deliver a therapeutic molecule while simultaneously. There are in literature several approaches to follow adcs for in vivo and in vitro studies because type of conjugation, release of the drug, and body distribution are characteristic for each of them, resulting in many critical steps. The commonly used bioanalytical methods typically implemented to characterize various adcs are summarized with a focus on the pharmacokinetic studies and determination of drug to antibody ratios. the challenges and perspectives of the assays are discussed as well. Detailing their journey from conception to becoming a leading treatment modality, we highlight the significant bioanalytical challenges and regulatory milestones that shape adc development. Antibody drug conjugates (adcs) represent a rapidly advancing class of biotherapeutics for oncology and immunological indications. comprehensive pharmacokinetic (pk) characterization is critical for assessing adcs efficacy, safety, and overall therapeutic performance.
Comments are closed.