Analysis Group Vac4eu

Analysis Group Vac4eu Ag provides rigorous methodological insights and data analysis to biopharmaceutical companies and healthcare organizations, supporting research across multiple therapeutic areas. Vac4eu developed a systematic, stepwise approach to validate vaccine safety outcomes. bc case definitions were utilized when available, adapted for rwd as needed, and newly developed for outcomes without an existing bc definition.
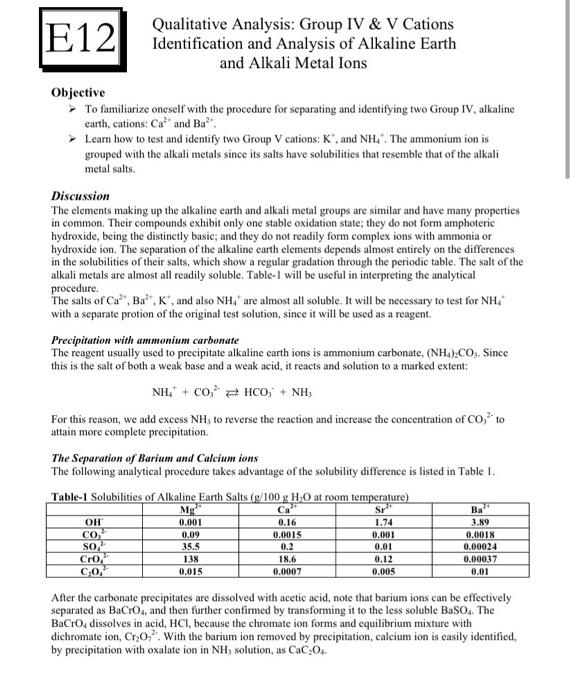
Solved E12 Qualitative Analysis Group Iv V Cations Chegg The vac4eu validation pipeline provides a standardized framework for training and validating vaccine safety outcome in rwd. by adapting bc case definitions and dedicated training of abstractors, we will reduce variability in outcome validation in post authorization safety studies. To address this, the vaccine monitoring collaboration for europe (vac4eu) developed a structured validation pipeline that operationalizes bc case definitions for vaccine safety outcomes in rwd in a harmonized, scalable, and reusable manner. Methods vac4eu developed a systematic, stepwise approach to validate vaccine safety outcomes. bc case definitions were utilized when available, adapted for rwd as needed, and newly developed for. The vaccine monitoring collaboration for europe (vac4eu) has developed a structured validation pipeline that operationalizes brighton collaboration (bc) case definitions for real world data (rwd) in a harmonized, scalable and reusable manner.
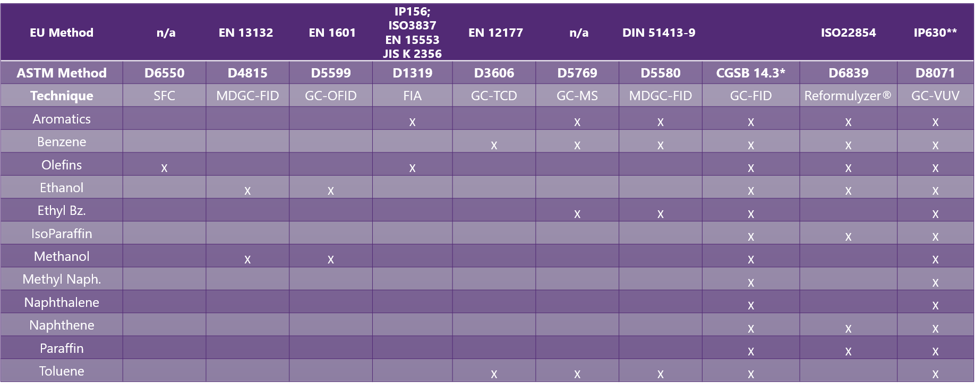
Vuv Analytics Methods vac4eu developed a systematic, stepwise approach to validate vaccine safety outcomes. bc case definitions were utilized when available, adapted for rwd as needed, and newly developed for. The vaccine monitoring collaboration for europe (vac4eu) has developed a structured validation pipeline that operationalizes brighton collaboration (bc) case definitions for real world data (rwd) in a harmonized, scalable and reusable manner. Valuation of the ad26.cov2.svaccine, including observational studies using real world data onthe useof the vaccine. to fulfil theseregulatory obligations, the sponsor initiated a post authorisation safety study (pass) to characterise and evaluate the safety profile of ad26.cov2.s in a large population sample size to inform the scien. The mission of vac4eu is to access, characterize, and analyze available and newly collected health data to allow for evidence based decisions by people who need to regulate, advise, prescribe, or decide on vaccines. We conduct collaborative post licensure studies on vaccine benefits, risk, and coverage utilising primary and secondary health data. we harmonise workflows through a quality management system, a common framework and shared tools including the conception common data model. The vaccine monitoring collaboration for europe (vac4eu) developed a validation pipeline designed to apply brighton collaboration (bc) case definitions adapted for rwd in a harmonized, reusable manner.
Comments are closed.