Alkenes
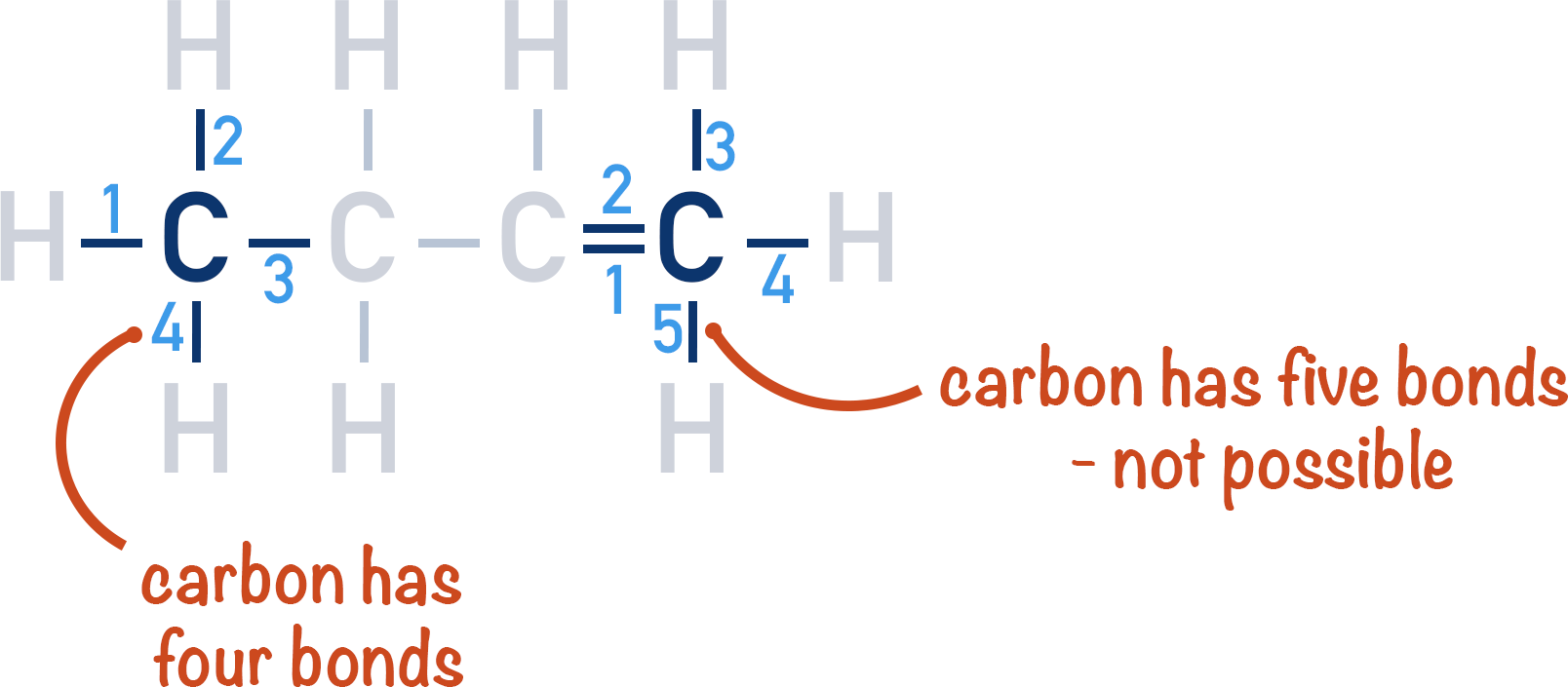
Alkenes A Level Chemistrystudent Alkenes are generally colorless non polar compounds, somewhat similar to alkanes but more reactive. the first few members of the series are gases or liquids at room temperature. This page covers alkenes, unsaturated hydrocarbons with carbon to carbon double bonds. it explains iupac naming conventions, emphasizing the longest carbon chain and the lowest numbering for double ….
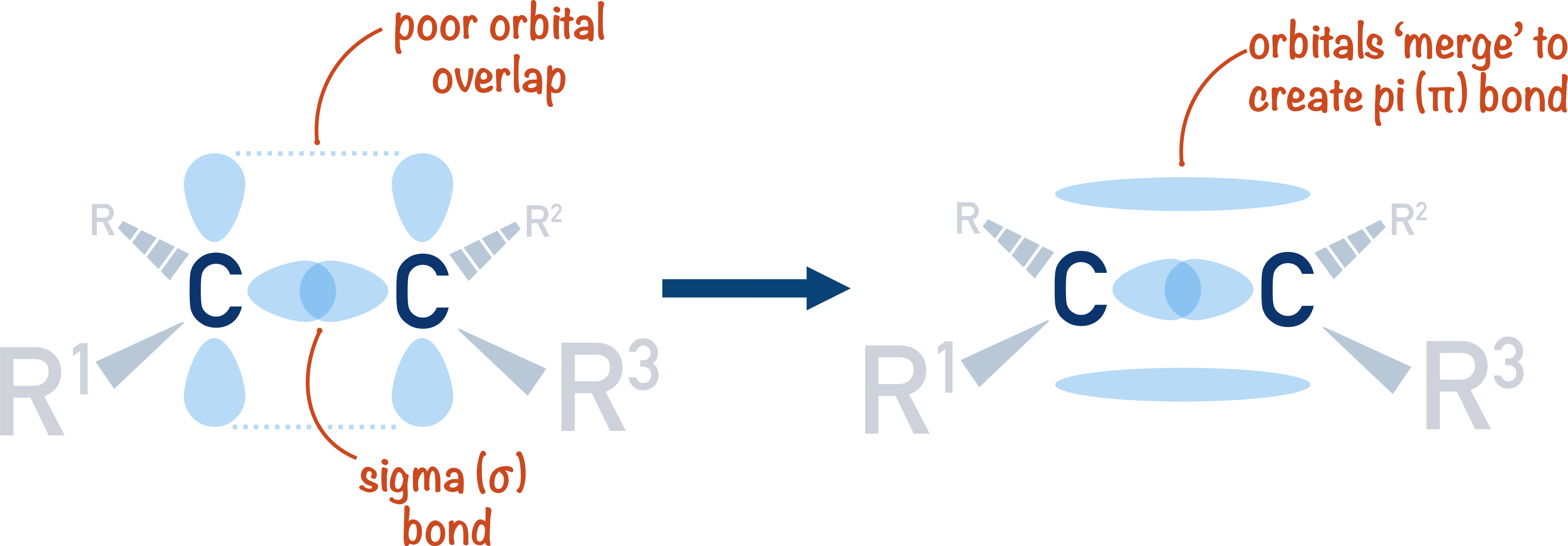
Alkenes A Level Chemistrystudent Learn about alkenes, unsaturated hydrocarbons with carbon carbon double bonds, and their nomenclature, isomerism, preparation, and properties. find out how alkenes react with different reagents and what are their applications in organic chemistry. Alkenes exhibit a trigonal planar shape around the double bond, with bond angles of approximately 120 degrees due to sp² hybridization of the carbon atoms. bonding and structure the double bond in alkenes consists of one sigma (σ) bond and one pi (π) bond, where the π bond is formed by the sideways overlap of p orbitals. Alkenes are hydrocarbons that contain at least one carbon carbon double bond. the general formula for alkenes is cnh2nc nh {2n}, where n represents the number of carbon atoms in the molecule. Alkenes are among the most common and important molecules in organic chemistry and biochemistry. in this article, you will learn the structure of alkene, its nomenclature, and some of its reactions.

Ch105 Chapter Alkenes Alkynes And Aromatic Compounds 42 Off Alkenes are hydrocarbons that contain at least one carbon carbon double bond. the general formula for alkenes is cnh2nc nh {2n}, where n represents the number of carbon atoms in the molecule. Alkenes are among the most common and important molecules in organic chemistry and biochemistry. in this article, you will learn the structure of alkene, its nomenclature, and some of its reactions. Explore the synthesis of alkenes through elimination reactions, focusing on e1 and e2 mechanisms and the role of strong bases in organic chemistry. Alkenes are the second group of hydrocarbons and differ from alkanes in that they have a double bond. the presence of a double bond brings up some important structural and functional changes. As noted before, alkenes are hydrocarbons with carbon to carbon double bonds (r 2 c=cr 2) and alkynes are hydrocarbons with carbon to carbon triple bonds (r–c≡c–r). Alkenes consist of carbon and hydrogen atoms, where at least two carbon atoms are linked by a double bond. this double bond makes them much more reactive than alkanes (single bonded hydrocarbons) and less saturated with hydrogen.

Branched Alkenes Explore the synthesis of alkenes through elimination reactions, focusing on e1 and e2 mechanisms and the role of strong bases in organic chemistry. Alkenes are the second group of hydrocarbons and differ from alkanes in that they have a double bond. the presence of a double bond brings up some important structural and functional changes. As noted before, alkenes are hydrocarbons with carbon to carbon double bonds (r 2 c=cr 2) and alkynes are hydrocarbons with carbon to carbon triple bonds (r–c≡c–r). Alkenes consist of carbon and hydrogen atoms, where at least two carbon atoms are linked by a double bond. this double bond makes them much more reactive than alkanes (single bonded hydrocarbons) and less saturated with hydrogen.

Naming Alkenes As noted before, alkenes are hydrocarbons with carbon to carbon double bonds (r 2 c=cr 2) and alkynes are hydrocarbons with carbon to carbon triple bonds (r–c≡c–r). Alkenes consist of carbon and hydrogen atoms, where at least two carbon atoms are linked by a double bond. this double bond makes them much more reactive than alkanes (single bonded hydrocarbons) and less saturated with hydrogen.
Comments are closed.