Alkene Reactions

Alkene Reactions Diagram Quizlet Alkenes are primarily prepared by elimination reactions of molecules that contain good leaving groups attached to sp 3 carbons. examples of such reactions are dehydrohalogenations with strong base, and acid catalyzed dehydrations of alcohols. the opposite of an elimination is an addition reaction. Summary of the reactions of alkenes into a reaction map, including reactions of alkanes and alkyl halides, containing 38 reactions.
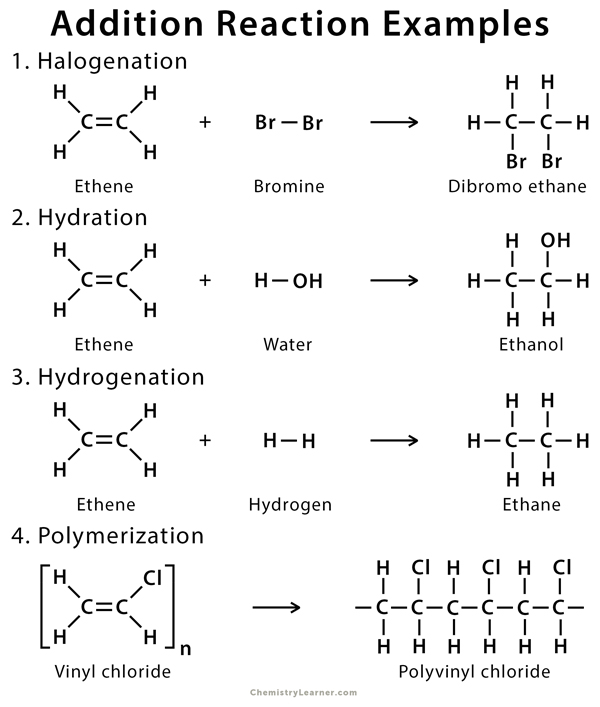
Alkene Reactions Definition Types And Mechanism Learn about the different types of alkene reactions, such as addition, elimination, and oxidation, and how they occur. find examples, formulas, and faqs on alkene reactions. The reaction involves the addition of ozone across the double bond of the alkene, resulting in the cleavage of the carbon carbon double bond. for symmetrical alkenes, two identical carbonyl compounds are formed, while for asymmetrical alkenes, a mixture of different carbonyl compounds is obtained. Electron donating double bond substituents increase the reactivity of an alkene, as evidenced by the increased rate of hydration of 2 methylpropene (two alkyl groups) compared with 1 butene (one alkyl group). Learn and review alkene reactions with this cheat sheet that summarizes products, reagents, regioselectivity and stereoselectivity. watch a video explaining the cheat sheet and practice with a quiz.
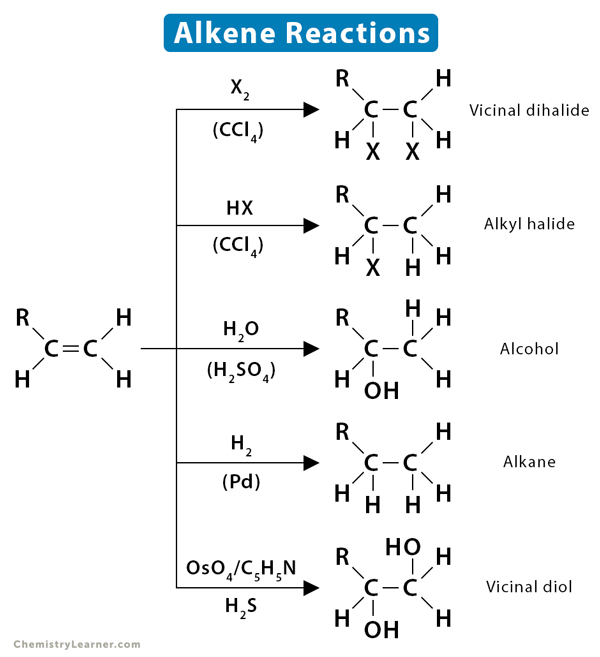
Alkene Reactions Definition Types And Mechanism Electron donating double bond substituents increase the reactivity of an alkene, as evidenced by the increased rate of hydration of 2 methylpropene (two alkyl groups) compared with 1 butene (one alkyl group). Learn and review alkene reactions with this cheat sheet that summarizes products, reagents, regioselectivity and stereoselectivity. watch a video explaining the cheat sheet and practice with a quiz. There are a ton of alkene reactions out there. we give you a useful chart of them here, along with our free study guide. This organic chemistry study guide covers alkene reactions: halogenation, hydration, reduction, oxidation, cyclopropane synthesis, and stereochemistry. Each bh 3 borane molecule can react with three alkene molecules. the reaction is regioselective, since boron is bonded to the least substituted carbon (the oh group is generated from the alkylborane by oxidation of the latter) and hydrogen to the most substituted carbon (anti markovnikov). A hydration reaction is a reaction in which water is added to an alkene. hydration reactions can take place when the alkene and water are heated to near 100 o c in the presence of a strong acid, which acts as a catalyst.
Comments are closed.