Alkene Halogenation %f0%9d%91%8e%f0%9d%91%9b%f0%9d%91%a1%f0%9d%91%96 Addition Bromoniumion Organic Reaction Mechanism 4k 006
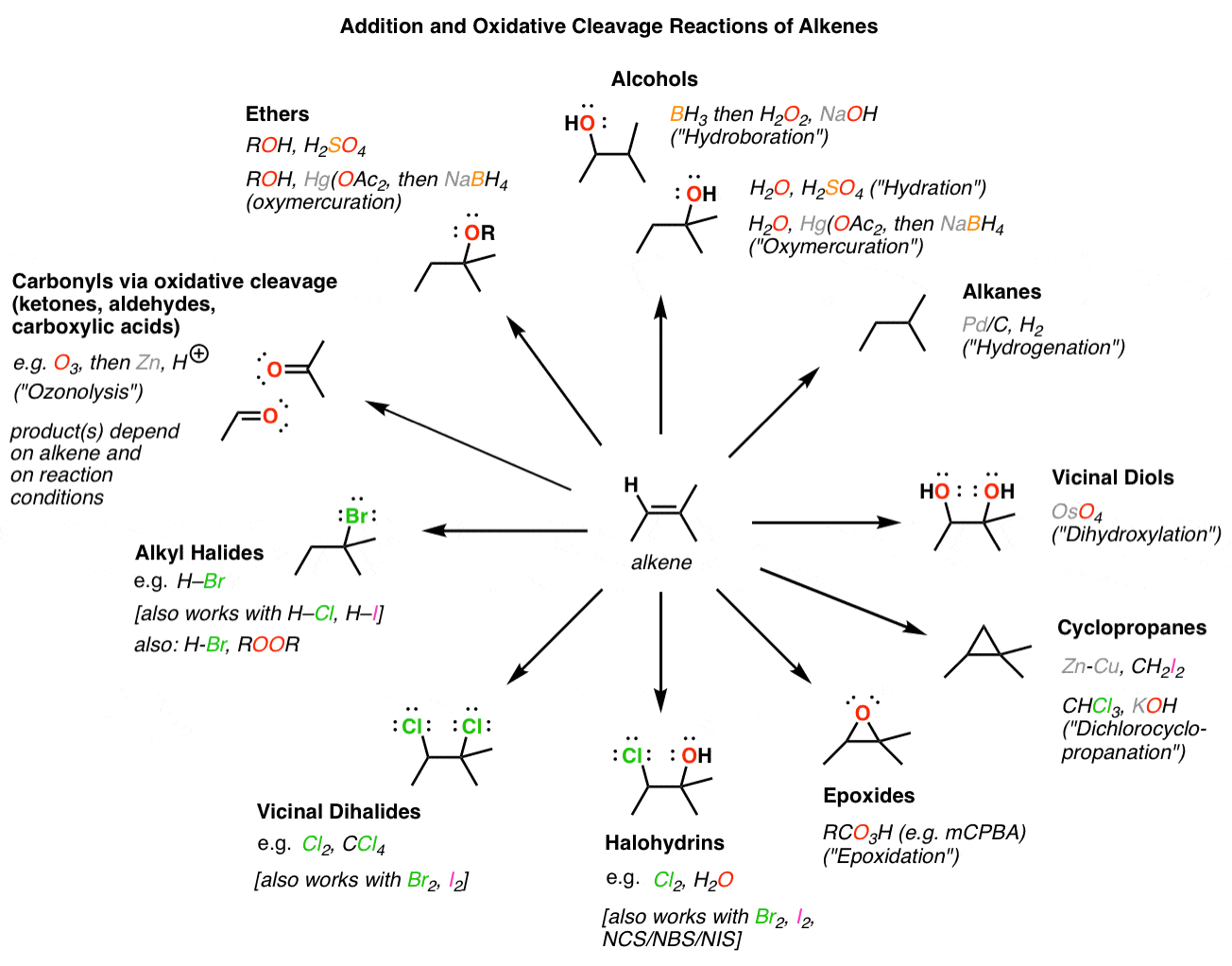
Alkene Addition Reaction Alkene Addition Sqmks Stereochemistry of this addition can be explained by the mechanism of the reaction. in the first step, the electrophilic halogen (with the positive charge) approaches the pi bond and 2p orbitals of the halogen bond with two carbon atoms creating a cyclic ion with a halogen as the intermediate. This reaction has a rather complicated mechanism, and it is also stereospecific, so the stereochemistry is always something to be very careful with. in this tutorial, we are going to cover everything you need to know about this reaction.

A Detailed Diagram Of An Organic Reaction Mechanism Premium Ai Halogenation of alkenes is an example of a stereoselective reaction. the reaction of br 2, cl 2 and other halogens with alkenes leads to products of anti – addition. Reaction overview: the alkene halogenation reaction, specifically bromination or chlorination, is one in which a dihalide such as cl2 or br2 is added to a molecule after breaking the carbon to carbon double bond. The reaction mechanism for an alkene bromination can be described as follows. in the first step of the reaction, a bromine molecule approaches the electron rich alkene carbon–carbon double bond. Bromine and chlorine add rapidly to alkenes to yield 1,2 dihalides, a process called halogenation. for example, nearly 50 million tons of 1,2 dichloroethane (ethylene dichloride) are synthesized worldwide each year, much of it by addition of cl 2 to ethylene.
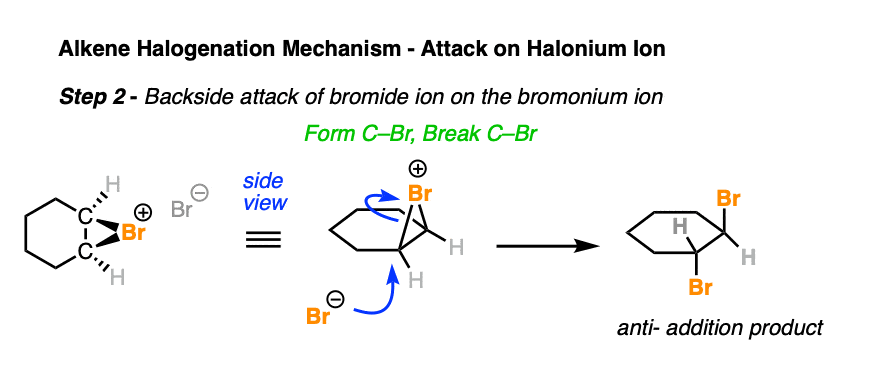
Bromination Of Alkenes The Mechanism Master Organic Chemistry The reaction mechanism for an alkene bromination can be described as follows. in the first step of the reaction, a bromine molecule approaches the electron rich alkene carbon–carbon double bond. Bromine and chlorine add rapidly to alkenes to yield 1,2 dihalides, a process called halogenation. for example, nearly 50 million tons of 1,2 dichloroethane (ethylene dichloride) are synthesized worldwide each year, much of it by addition of cl 2 to ethylene. The halogens br 2 and cl 2 add to the double bond of an alkene, producing vicinal dihalides – a compound bearing the halogens on adjacent carbons (vicinus, latin: adjacent). these are also called 1,2 dihalides: the r eaction with bromine is a standard test for the presence of a π bond. Draw the structure of the product formed when a given alkene undergoes an addition reaction with chlorine or bromine. write the mechanism for the addition reaction that occurs between an alkene and chlorine or bromine, and account for the stereochemistry of the product. A halogenation addition reaction occurs between halogens (br 2 and cl 2) and alkenes, creating two adjacent cx bonds, where x is a halogen (br or cl). for example, bromination adds one bromine atom to each alkene carbon, resulting in a dibromoalkane. Master the halogenation of an alkene. discover the cyclic halonium ion mechanism, anti addition stereochemistry, and industrial uses in this expert guide.
Comments are closed.