Alkane Alkene Pdf Alkane Alkene

Alkane Alkene Alkyne Pdf Download Free Pdf Alkane Alkene Alkane, alkene, alkyne.pdf free download as pdf file (.pdf), text file (.txt) or read online for free. the document summarizes the key properties and reactions of alkanes, alkenes, and alkynes. The alkane molecules break up to form a smaller alkane molecule and at the same time an alkene, such as ethene, is formed which can then be used to make polymers.

Alkane Alkene Alkyne S2 Pdf Ethene, c2h4, is the simplest alkene. each carbon atom in ethene, commonly called ethylene, has a trigonal planar structure. In this section, we will focus on the structure of the long fatty acid tails, which can be composed of alkane or alkene structures. chapter 10 will focus more on the formation of the ester bonds. In the following example, two different chains in the same alkane have seven c atoms. we circle the longest continuous chain as shown in the diagram on the left, since this results in the greater number of substituents. The name of an alkene is derived from the name of the alkane with the same number of carbon atoms. the presence of the double bond is signified by replacing the suffix ane with the suffix ene.

Alkane Alkene Alkyne Pdf In the following example, two different chains in the same alkane have seven c atoms. we circle the longest continuous chain as shown in the diagram on the left, since this results in the greater number of substituents. The name of an alkene is derived from the name of the alkane with the same number of carbon atoms. the presence of the double bond is signified by replacing the suffix ane with the suffix ene. Alkanes are saturated hydrocarbons characterized by single carbon carbon bonds, while alkenes contain carbon carbon double bonds. various synthesis techniques and reactions, such as hydrogenation, dehydrohalogenation, and purification methods are discussed, along with applications of these compounds. alkene (organic chem) (1).pptx . 13. Unlike the complex transformations of combustion, the halogenation of an alkane appears to be a simple substitution reaction in which a c h bond is broken and a new c x bond is formed. Alkane, alkene, alkyne naming naming straight chain alkanes: find the longest continuous chain of c atoms. choose the appropriate prefix. the name ends with –ane. Some alkene or alkenyl substituents have common names. the simplest alkene, ch2=ch 2, named in the iupac system as ethene, is often called ethylene.
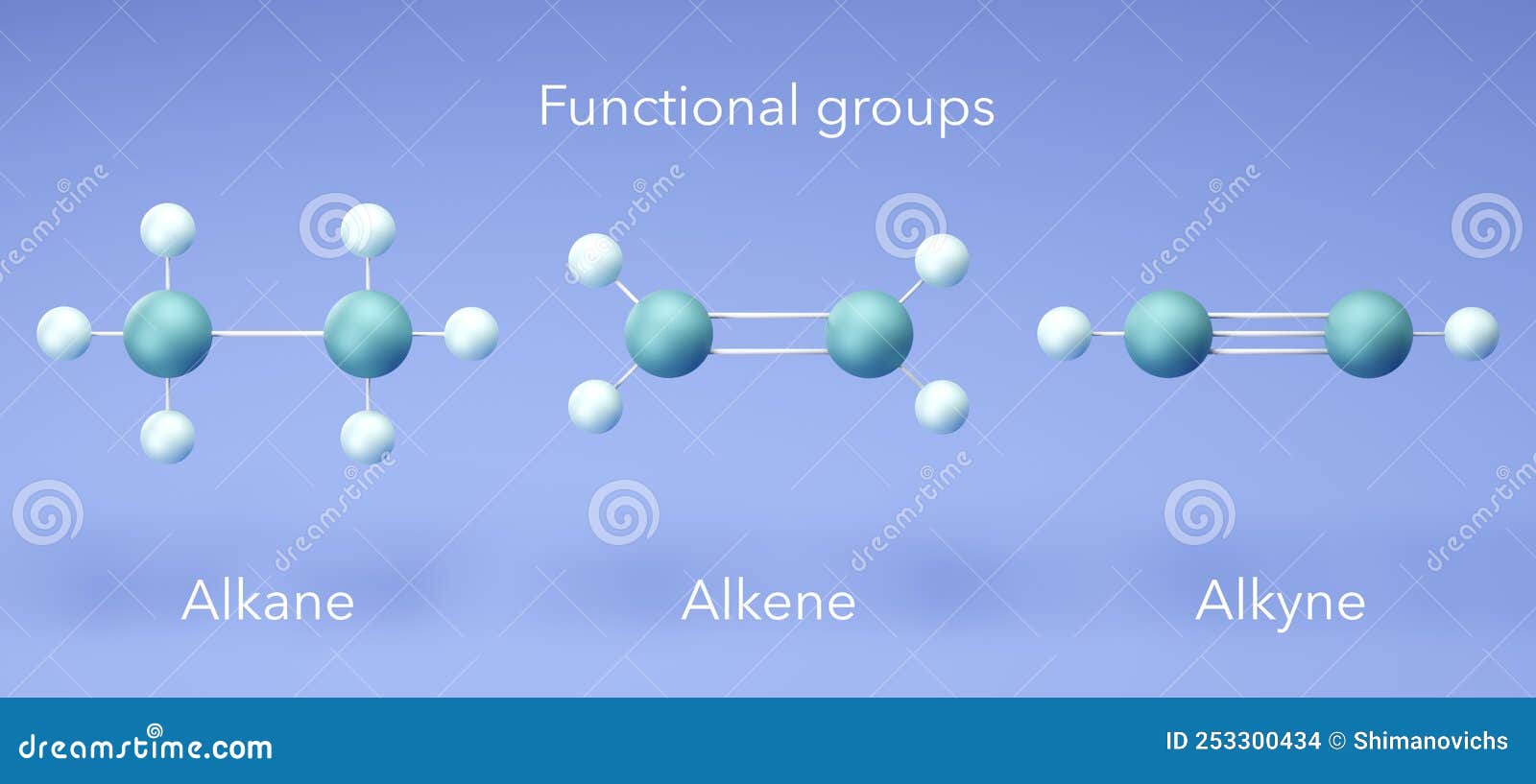
Alkane Alkene Alkyne Structure My Xxx Hot Girl Alkanes are saturated hydrocarbons characterized by single carbon carbon bonds, while alkenes contain carbon carbon double bonds. various synthesis techniques and reactions, such as hydrogenation, dehydrohalogenation, and purification methods are discussed, along with applications of these compounds. alkene (organic chem) (1).pptx . 13. Unlike the complex transformations of combustion, the halogenation of an alkane appears to be a simple substitution reaction in which a c h bond is broken and a new c x bond is formed. Alkane, alkene, alkyne naming naming straight chain alkanes: find the longest continuous chain of c atoms. choose the appropriate prefix. the name ends with –ane. Some alkene or alkenyl substituents have common names. the simplest alkene, ch2=ch 2, named in the iupac system as ethene, is often called ethylene.
Comments are closed.