Alcohol Functional Group Examples

Alcohol Functional Group Examples An alcohol is an organic compound with a hydroxyl (oh) functional group on an aliphatic carbon atom. because oh is the functional group of all alcohols, we often represent alcohols by the general formula roh, where r is an alkyl group. alcohols are common in nature. Methanol (or methyl alcohol or wood alcohol; ch 3 oh) is most commonly used to make fuels for cars and ships. ethanol (or ethyl alcohol; c 2 h 5 oh) is the alcohol found in alcoholic drinks.
Alcohol Functional Group Examples Alcohols are derivatives of hydrocarbons in which an –oh group has replaced a hydrogen atom. although all alcohols have one or more hydroxyl (–oh) functional groups, they do not behave like bases such as naoh and koh. naoh and koh are ionic compounds that contain oh – ions. Alcohols are organic molecules containing the “hydroxyl” functional group, “oh” directly bonded to carbon. the carbon directly attached to oh is technically called the “carbinol” carbon, although this nomenclature is often not introduced in introductory classes. In chemistry, an alcohol (from arabic al kuḥl 'the kohl ') [2] is a type of organic compound that carries at least one hydroxyl (−oh) functional group bound to a saturated carbon atom. [3][4] alcohols range from the simple, like methanol and ethanol, to complex, like sugar alcohols and cholesterol. Learn about the alcohol functional group. see primary and secondary alcohol examples and find out how to classify types of alcohol. learn the difference between alcohols and alkanols.
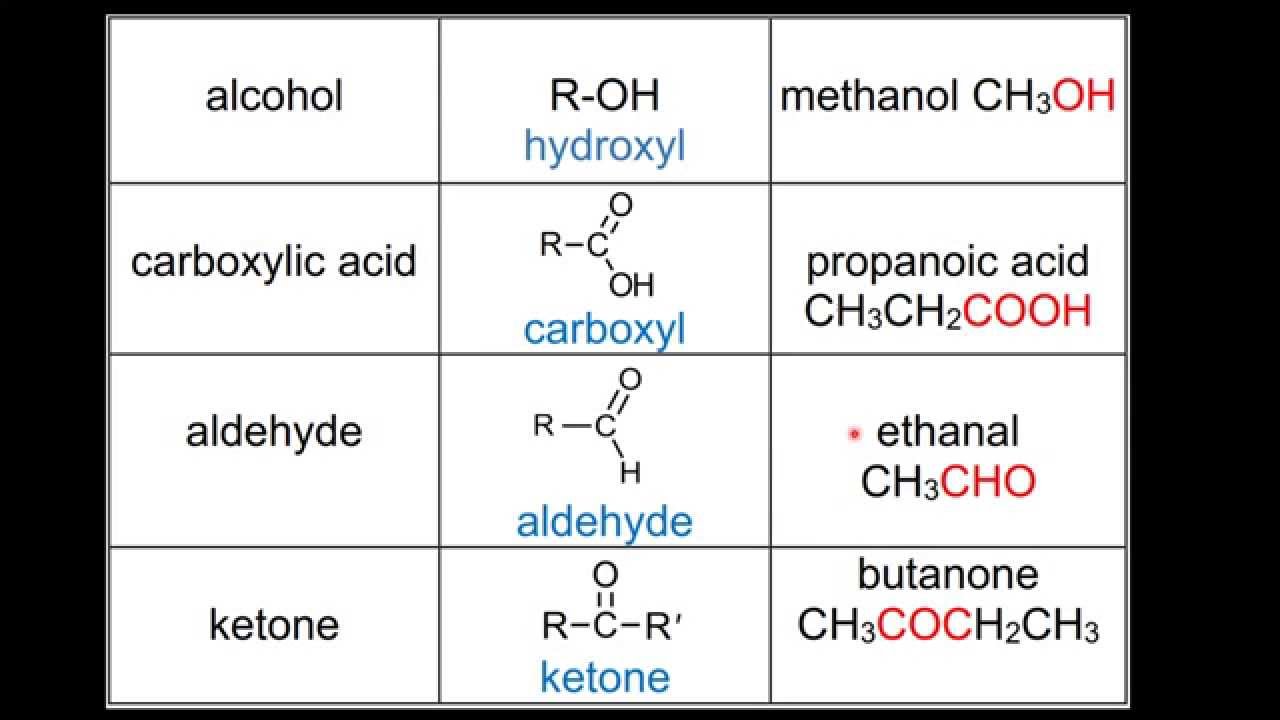
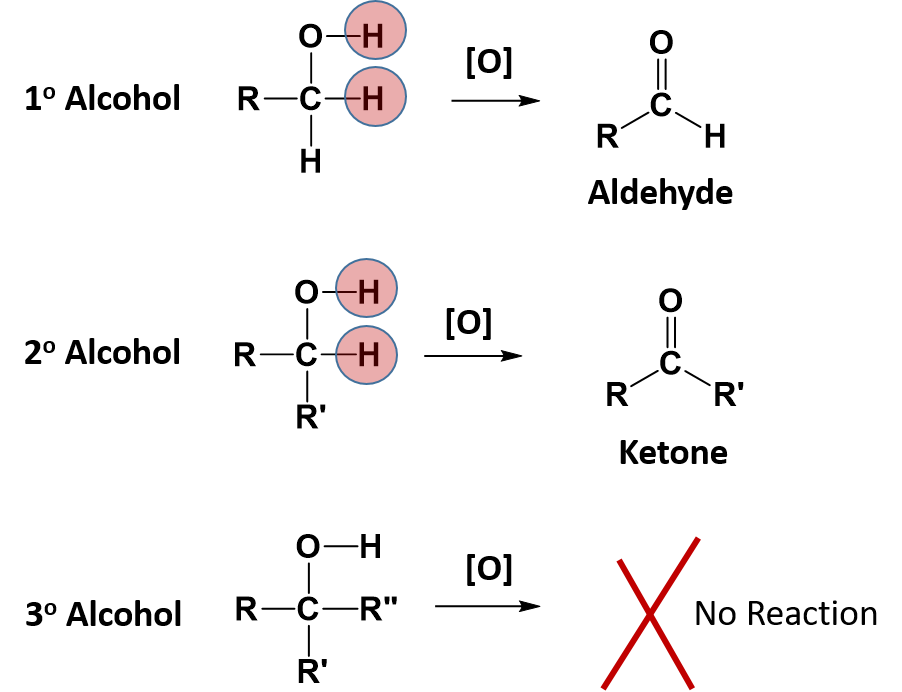
Alcohol Functional Group Examples In chemistry, an alcohol (from arabic al kuḥl 'the kohl ') [2] is a type of organic compound that carries at least one hydroxyl (−oh) functional group bound to a saturated carbon atom. [3][4] alcohols range from the simple, like methanol and ethanol, to complex, like sugar alcohols and cholesterol. Learn about the alcohol functional group. see primary and secondary alcohol examples and find out how to classify types of alcohol. learn the difference between alcohols and alkanols. Most people are familiar with ethyl alcohol (ethanol), the active ingredient in alcoholic beverages, but this compound is only one of a family of organic compounds known as alcohols. Alcohols can be classified as primary, secondary or tertiary alcohols. this classification relies on the alpha carbon and the number of hydrogen atoms bonded directly to the alpha carbon. Ethanol and resveratrol, a phenol, are representatives of two of the families of oxygen containing compounds that we consider in this chapter. two other classes, aldehydes and ketones, are formed by the oxidation of alcohols. ethers, another class, are made by the dehydration of alcohols. An alcohol is an organic compound with a hydroxyl (oh) functional group on an aliphatic carbon atom. because oh is the functional group of all alcohols, we often represent alcohols by the general formula roh, where r is an alkyl group.
Comments are closed.