Adaptive Clinical Trials Pptx
Adaptive Clinical Trials Global Events Usa Europe Middle East The fda has provided guidance on adaptive designs to ensure patient safety and that adaptive trials meet evidentiary standards for approval. download as a pptx, pdf or view online for free. Adaptive platform and umbrella protocols offer several practicable and desirable benefits which facilitate speedier answers and allow clinical trials to serve as a tool to move treatment on for patients much more quickly.

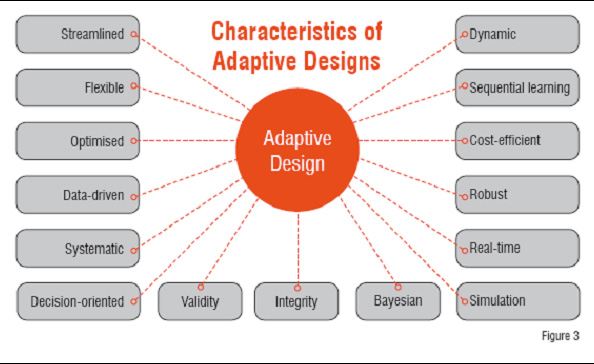
Adaptive Designs In Clinical Trials Rationale Execution Reporting Adaptive designs in clinical trials by dr pooja awasthi, department of pharmacology and therapeutics ,kgmu, lucknow free download as powerpoint presentation (.ppt .pptx), pdf file (.pdf), text file (.txt) or view presentation slides online. Learn about adaptive designs in clinical trials, including motivations, examples, and challenges faced in implementation. discover how adaptive features improve ethicality, safety, and economic aspects of trials with practical considerations discussed. Adaptive clinical trials allow for modifications to trial design based on data collected during the study, enhancing flexibility and efficiency without compromising integrity. The title describes a novel clinical trial design with several key features, so i will describe those, and present the results of the study we performed to test their utility.
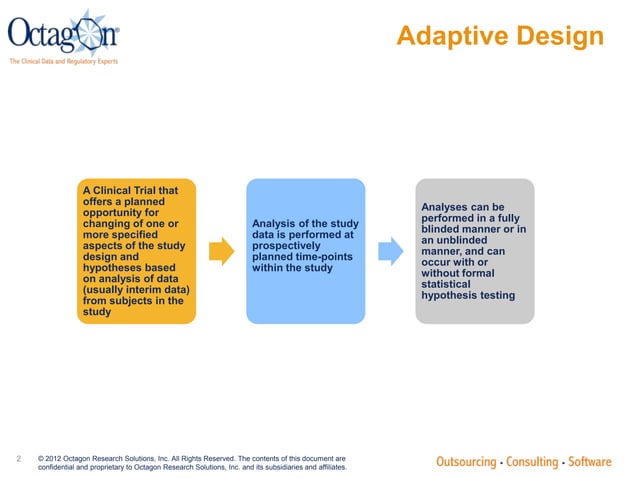
Adaptive Clinical Trials Pptx Adaptive clinical trials allow for modifications to trial design based on data collected during the study, enhancing flexibility and efficiency without compromising integrity. The title describes a novel clinical trial design with several key features, so i will describe those, and present the results of the study we performed to test their utility. Focuses on key principles in design, conduct, analysis, and reporting. expands discussion on technical aspects such as estimation, simulations, bayesian methods. adds clarity on what should be submitted. regulatory principles for adaptive designs. chance of erroneous conclusions should be adequately controlled. Download presentation by click this link. while downloading, if for some reason you are not able to download a presentation, the publisher may have deleted the file from their server. The talk highlights the scientific, ethical, and operational advantages of bayesian designs, along with challenges such as regulatory scrutiny and computational demands. it concludes with future prospects involving ai and decentralized trial models. download as a pptx, pdf or view online for free. Pushpakom et al. (2015), “telmisartan and insulin resistance in hiv (tailor): protocol for a dose ranging phase ii randomised open labelled trial of telmisartan as a strategy for the reduction of insulin resistance in hiv positive individuals on combination antiretroviral therapy.”,.
Comments are closed.