Acids Bases And Buffers

Acids Bases And Buffers Download Free Pdf Buffer Solution Acid A solution of acetic acid (ch a 3 cooh and sodium acetate ch a 3 coona) is an example of a buffer that consists of a weak acid and its salt. an example of a buffer that consists of a weak base and its salt is a solution of ammonia (nh a 3 (aq)) and ammonium chloride (nh a 4 cl (aq)). Define acids and bases with examples using different theories. define neutralization reaction with example. explain the limitations of arrhenius theory. define buffer, buffer capacity.
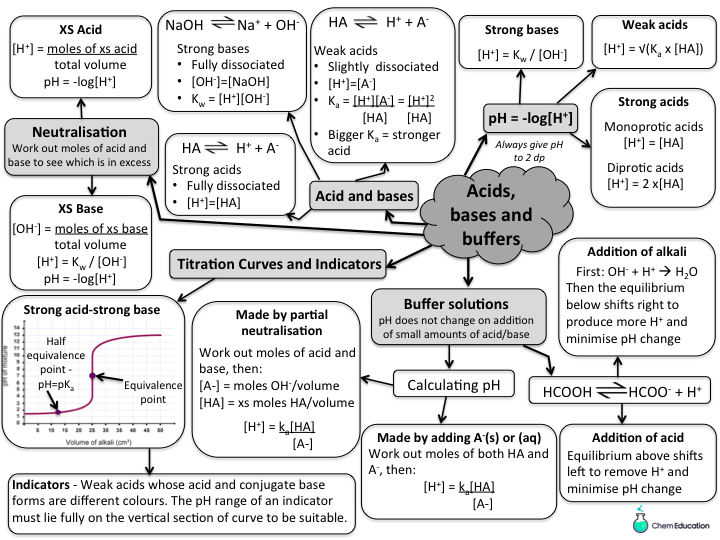
Acids Bases And Buffers Revision Bundle Teaching Resources Describe the composition and function of acid–base buffers. calculate the ph of a buffer before and after the addition of added acid or base. The salt content can be added in several ways: a salt solution could be added to the acid or some solid salt added. a buffer can also be made by partially neutralising a weak acid with alkali and therefore producing a mixture of salt and acid. This study guide covers acids, bases, ph, titrations, buffers, equilibrium, and molecular structure, helping you master general chemistry unit 8. Acids are substances that provide hydrogen ions (h ) and lower ph, whereas bases provide hydroxide ions (oh –) and raise ph. the stronger the acid, the more readily it donates h . for example, hydrochloric acid and lemon juice are very acidic and readily give up h when added to water.

Solution Acids Bases And Buffers Comprehensive Module Studypool This study guide covers acids, bases, ph, titrations, buffers, equilibrium, and molecular structure, helping you master general chemistry unit 8. Acids are substances that provide hydrogen ions (h ) and lower ph, whereas bases provide hydroxide ions (oh –) and raise ph. the stronger the acid, the more readily it donates h . for example, hydrochloric acid and lemon juice are very acidic and readily give up h when added to water. Buffer solutions are aqueous solutions that resist significant changes in ph when small amounts of an acid or base are added. buffers are crucial in maintaining stable ph levels in various chemical, biological, and industrial systems. they are typically composed of a weak acid and its conjugate base, or a weak base and its conjugate acid. Stomach acid is hydrochloric acid required for the digestion of food. a base is commonly referred to as any substance that can neutralize an acid, feel slippery to the touch when in solution, and taste bitter. a brønsted lowry acid is a proton donor, while a brønsted lowry base is a proton acceptor. Acid base chemistry: how do organisms maintain a constant ph range? acidity and basicity, proton concentration, the ph scale, and buffers. Ap chem guide's crash course on acid base reactions and buffers.
Comments are closed.