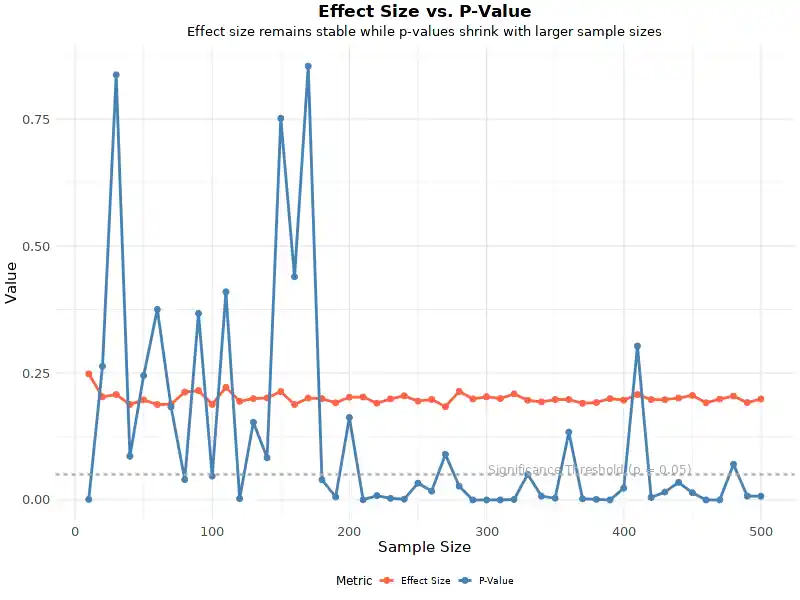
A Graph Of P Rp Vs P Rs Vs P Ps Shows A Trajectory In Which All
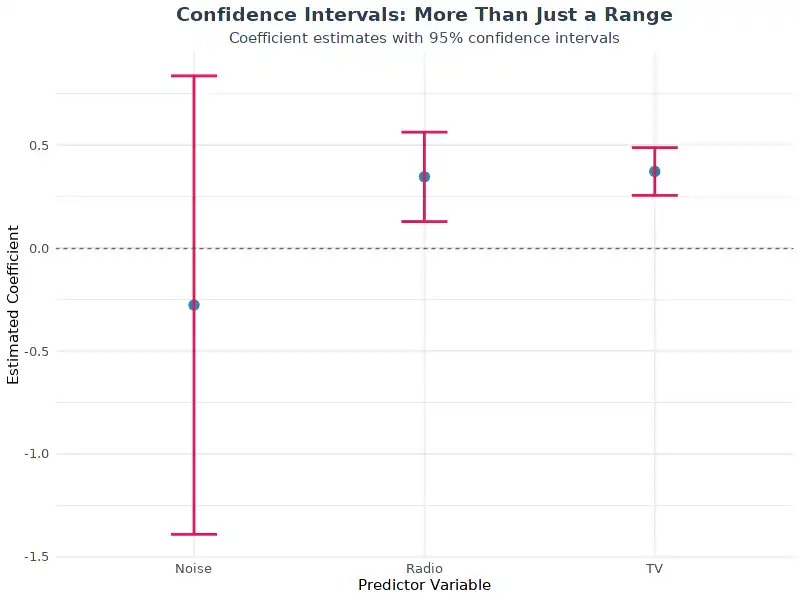
Redirecting A graph of p [rp] vs. p [rs] vs. p [ps] shows a trajectory in which all three variables start at 0.1 and converge at the same rate, hence the linear graph, and they all. Every point on a pv diagram represents a different state for the gas (one for every possible volume and pressure). as a gas goes through a thermodynamics process, the state of the gas will shift around in the pv diagram, tracing out a path as it moves (as shown in the diagram below).
Redirecting Include a sign with all numbers associated with a path. the pressure volume, and temperature numbers are always positive, so adding a sign to them would be redundant. On the figure we show two types of plots that are used to describe changes of state. on the left we have plotted the pressure versus the volume, which is called a p v diagram. These diagrams allow engineers and scientists to visualize the relationships between pressure, volume, and temperature, and can provide important insights into the efficiency and performance of engines, refrigeration systems, and other thermodynamic processes. To fully conceptualize consumer surplus, take an example of a demand curve of chocolates plotted on a graph. the unit price is plotted on the y axis and the actual chocolate units of demand per day on the x units.

A Graph Of P Rp Vs P Rs Vs P Ps Shows A Trajectory In Which All These diagrams allow engineers and scientists to visualize the relationships between pressure, volume, and temperature, and can provide important insights into the efficiency and performance of engines, refrigeration systems, and other thermodynamic processes. To fully conceptualize consumer surplus, take an example of a demand curve of chocolates plotted on a graph. the unit price is plotted on the y axis and the actual chocolate units of demand per day on the x units. You will remember that in a competitive market, the demand curve is flat. its slope is zero. so, the derivative of this curve, which is the mr curve, also has a slope of zero (two times zero = zero). so the result that p (max) occurs when mr = mc is true not just in a monopoly, but all markets. In conclusion, the agreement of the data with boyle's law can be represented in three different ways. a plot of pressure versus volume shows that as volume increases, pressure decreases (and vice versa), indicating an inverse relationship. Pressure volume (p v) diagrams are pivotal in the field of thermodynamics, offering a graphical representation of the changes in pressure and volume in a thermodynamic system. The somewhat triangular area labeled by f in the graph shows the area of consumer surplus, which shows that the equilibrium price in the market was less than what many of the consumers were willing to pay.
Comments are closed.